April 2026

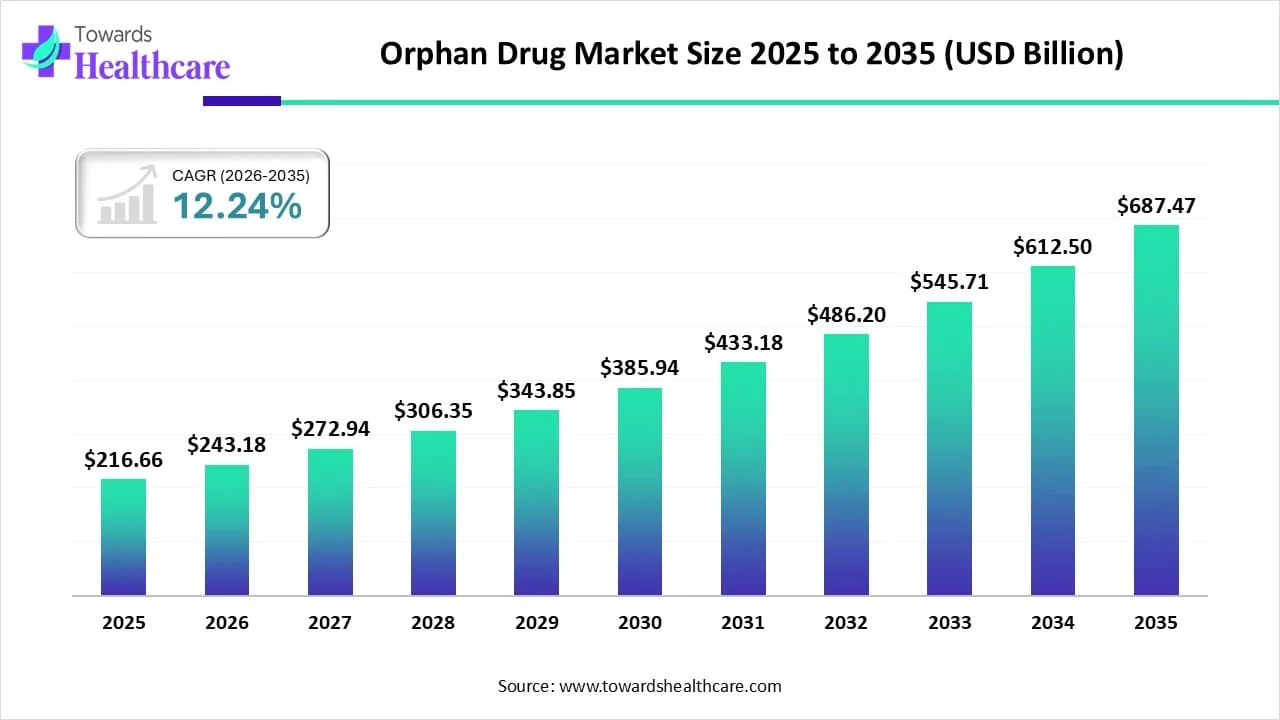
The global orphan drug market size is calculated at USD 216.66 billion in 2025, grew to USD 243.18 billion in 2026, and is projected to reach around USD 687.47 billion by 2035. The market is expanding at a CAGR of 12.24% between 2026 and 2035. The market is fueled by supportive regulations, advancements in genomics and personalized medicine, increased awareness of rare diseases, patient advocacy, financial incentives, and growing pharmaceutical investments in rare disease treatments.
Orphan drugs are medications designed to treat rare diseases that, while affecting a small number of people, often result in life-threatening or long-term debilitating conditions. As the prevalence of rare diseases and certain rare cancers rises, the demand for orphan drugs has surged. Many industry players are focusing on developing orphan drugs for the diagnosis, prevention, and treatment of these conditions. The USFDA also provides orphan drug designation, offering flexibility in production, which has encouraged strategic initiatives in the field. A recent report from the National Center for Advancing Translational Sciences revealed that almost 10,000 rare diseases impact the U.S. population. Additionally, the International Agency for Research on Cancer (IARC) states that around 25% of all cancer diagnoses are related to rare cancers. Due to the increasing approval of therapeutic drugs, many industry players have ramped up the development of orphan drugs, including biologics, to treat patients with rare diseases.
AI is playing a pivotal role in revolutionizing the market by accelerating drug discovery, enhancing clinical trial design, and improving patient recruitment. Machine learning algorithms can analyze vast datasets, identify potential drug candidates, and predict how rare diseases may respond to specific treatments. AI-driven platforms help pharmaceutical companies prioritize compounds with the highest success rates, thereby reducing development timelines and costs. Furthermore, AI is instrumental in personalized medicine by analyzing genetic data to develop targeted therapies for rare diseases, ensuring treatments are more effective and tailored to individual patient needs. This integration of AI is driving innovation and significantly advancing the development of orphan drugs.
How is the orphan oncology space expected to evolve globally over the next decade?
What payer and reimbursement dynamics should rare oncology drug developers anticipate?
How will digital health and data analytics influence orphan oncology drug development?
Orphan oncology is rapidly emerging as one of the most significant segments within the broader orphan drugs industry. The increasing focus on rare cancers, advancements in precision medicine, and the growing availability of targeted therapies have accelerated research and development in this area. Pharmaceutical companies are prioritizing orphan oncology because rare cancer indications often qualify for regulatory incentives such as market exclusivity, tax credits, and accelerated approval pathways. These benefits reduce development risks while addressing critical unmet medical needs.
Additionally, innovations in genomics and biomarker-based therapies are enabling more precise diagnosis and treatment of rare cancers, further strengthening the pipeline of orphan oncology drugs. As healthcare systems and regulators continue to support rare disease treatments, orphan oncology is expected to remain a key driver of growth in the orphan drugs market, attracting significant investment and expanding therapeutic options for patients with rare and underserved cancer types.
Driver
The rising prevalence of rare diseases is a key driver for the orphan drug market because it increases the demand for targeted therapies to treat conditions that affect a small portion of the population. As more patients are diagnosed with rare diseases, the need for specialized treatments grows, prompting pharmaceutical companies to invest in developing orphan drugs. Additionally, the growing awareness and advancements in diagnostics have led to more accurate identification of rare diseases, further fueling the demand for effective treatment options. In September 2024, Novo Nordisk partnered with NanoVation Therapeutics to advance genetic medicines for rare and cardiometabolic diseases, leveraging NanoVation’s proprietary long-circulating lipid nanoparticle technology.
Restraint
Orphan drugs are expensive because they target small patient populations, resulting in limited market size and higher development and manufacturing costs. Strict regulatory requirements, while ensuring safety and efficacy, lead to lengthy approval processes and additional compliance expenses. Navigating varying global regulations also adds complexity, further limiting the orphan drug market growth. In the United States, the average yearly cost of orphan drugs is about $32,000, with more than a third of these drugs exceeding $100,000 annually. For instance, enzyme replacement therapies can cost over $150,000 per year of treatment.
Opportunity
The growing focus on personalized therapeutics offers a significant opportunity for the orphan drug market. As genetic research advances, treatments can be more precisely tailored to rare diseases, improving their effectiveness. This shift towards precision medicine not only increases the demand for targeted therapies but also encourages more investment in orphan drug development, expanding both market potential and treatment options for patients with rare conditions. For example, in February 2024, Rapid Commercialization Partners and Orphan entered into a strategic collaboration to develop and market orphan drugs and therapies. In addition to industry stakeholders, various government organizations are also actively launching initiatives to support orphan drug research.
| Key Elements | Scope |
| Market Size in 2025 | USD 216.66 Billion |
| Projected Market Size in 2035 | USD 687.47 Billion |
| CAGR (2026 - 2035) | 12.24% |
| Leading Region | North America |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Drug Type, By Therapy, By Distribution Channel and By Region |
| Top Key Players | F. Hoffmann-La Roche Ltd., Celgene Corporation, Merk & Co., Inc., Novartis AG, Sanofi, Takeda Pharmaceutical Company Limited |
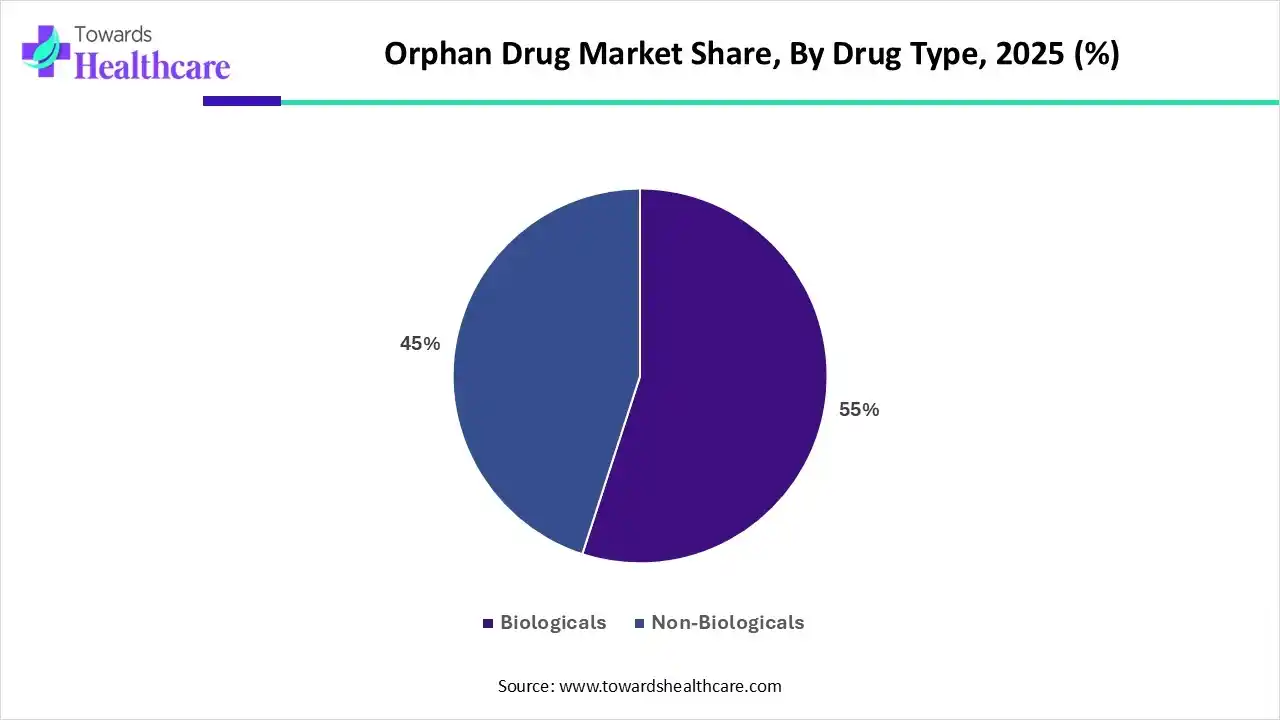
| Segments | Shares% |
| Biologicals | 55% |
| Non-Biologicals | 45% |
Explanation
Which Drug Type Segment Dominated the Orphan Drug Market?
By drug type, the biological segment held a dominant presence in the market in 2025. This segment dominated because biological drugs, which include monoclonal antibodies, gene therapies, and enzyme replacement therapies, are particularly well-suited for rare conditions with complex genetic underpinnings. These treatments often target the root causes of diseases at a molecular level, offering higher precision and better outcomes compared to traditional small-molecule drugs. Additionally, advancements in biotechnology and growing research into rare diseases have led to a surge in the development of biologics, driving their dominance in the orphan drug market.
The Non-biological Segment: Significantly Growing
By drug type, the non-biological segment is expected to grow at a significant rate in the coming years. Non-biological drugs, which include small molecules and oral medications, are often easier and more cost-effective to manufacture compared to biologics. With advancements in drug delivery systems and formulation technologies, these non-biological treatments are becoming more effective for managing rare diseases. Additionally, as the demand for affordable orphan drugs rises, non-biological therapies present a promising alternative to high-cost biologics, making them increasingly attractive for both pharmaceutical companies and healthcare systems. Their potential for broad application across various rare diseases and the growing emphasis on cost-efficiency in healthcare further contribute to the expected growth of this segment.
| Segments | Shares% |
| Oncology | 35% |
| Hematology | 10% |
| Neurology | 10% |
| Infectious Diseases | 10% |
| Metabolic Disorders | 10% |
| Endocrinology | 5% |
| Immunology | 5% |
| Others | 15% |
Explanation
Why Did the Oncology Segment Dominate the Orphan Drug Market?
By therapy, the oncology segment held the largest share of the market by 35% in 2025. This is due to oncology has seen a rapid rise in the development of orphan drugs, particularly as more targeted therapies and immunotherapies have emerged to treat rare and difficult-to-treat cancers. Many of these cancers have genetic or molecular characteristics that make them suitable for orphan drug treatments, which are tailored to the specific needs of small patient populations. Additionally, cancer research has received substantial funding, and rare cancers often lack effective treatment options, driving the demand for new therapies. The combination of unmet medical needs, targeted treatment approaches, and ongoing scientific progress has led to oncology's dominant presence in the orphan drug market.
The Neurology Segment: Fastest-growing
By therapy, the neurology segment is estimated to grow at the fastest rate in the market during the forecast period. Neurological disorders, many of which are rare and genetically driven, have limited treatment options, creating a strong demand for specialized orphan drugs. Moreover, as the understanding of the genetic and molecular causes of neurological disorders improves, drug development becomes more focused and effective, accelerating the introduction of new therapies. Additionally, increasing investment and research in the neurology field, along with a growing awareness of rare neurological conditions, further contribute to the rapid growth of this segment in the orphan drug market.
| Segment | Share 2025 (%) |
| Hospital Pharmacies | 50% |
| Retail Pharmacies | 20% |
| Online Pharmacies | 20% |
| Others | 10% |
Explanation
How the Hospital Pharmacies Segment Dominated the Orphan Drug Market?
By distribution, the hospital pharmacies segment was dominant in the market by 50% in 2025. Orphan drugs are typically administered in hospital settings where healthcare professionals can closely monitor patient responses and manage potential side effects. Hospital pharmacies also provide the infrastructure and expertise needed for preparing and dispensing these high-cost, specialized treatments. Additionally, many orphan drugs, especially biologics, need to be stored and handled under strict conditions, further driving their distribution through hospital pharmacies, which are equipped to manage these requirements.
The Online Pharmacies Segment: Significant CAGR
By distribution, the online pharmacies segment is estimated to grow at a significant CAGR during the predicted timeframe due to increased accessibility, convenience, and evolving regulatory support. These platforms allow patients, especially those in remote areas, to easily access rare and expensive medications. Additionally, online pharmacies offer competitive pricing, patient support services, and faster delivery, which enhances treatment adherence. The rise of telemedicine, digital health platforms, and consumer preference for privacy further boosts the demand, making online pharmacies a key distribution channel for orphan drugs.
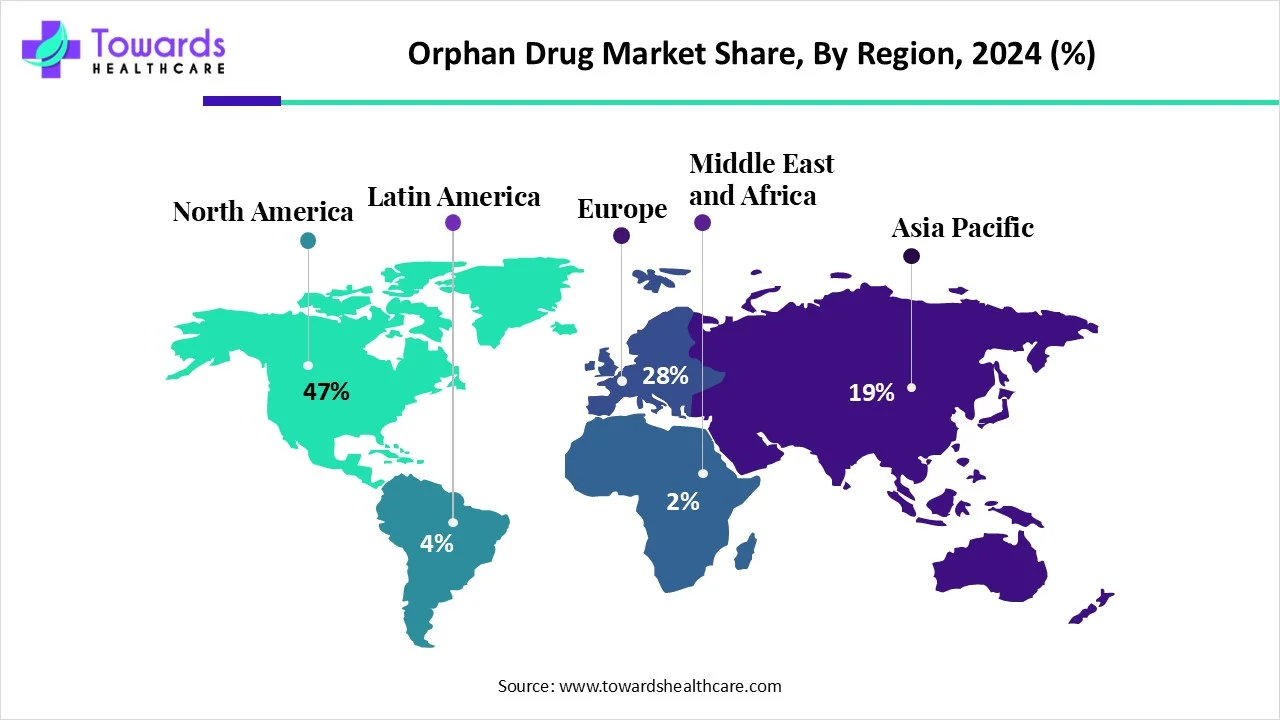
North America led the orphan drug market share by 47% in 2025. First, the region has a well-established healthcare infrastructure and a high level of awareness about rare diseases, which has driven the demand for orphan drugs. The region has a large patient population and significant investment in healthcare innovation, making it an attractive market for both pharmaceutical companies and biotech firms. The region also benefits from robust healthcare insurance coverage, which helps patients access costly orphan drugs. Furthermore, the growing adoption of telemedicine and digital health solutions in North America has facilitated easier access to these specialized treatments. These factors collectively position North America as a dominant force in the orphan drug market.
The U.S. Orphan Drug Market Trend
The U.S. market grows due to incentives from the Orphan Drug Act, which offers tax credits, grants, and market exclusivity for rare disease treatments. A large patient population, advanced healthcare infrastructure, and strong regulatory support, including faster FDA approvals, also drive growth. Additionally, increasing awareness, biotech innovation, and insurance coverage help make orphan drugs more accessible to patients. The market has also become dominant as rare diseases have jumped significantly in the United States.
The Canada Orphan Drug Market Trends
The Canadian market grows due to government support through initiatives like the Orphan Drug Program, a well-established healthcare system, and increasing awareness of rare diseases. Regulatory support from Health Canada, along with partnerships and patient assistance programs, also drive demand and make orphan drugs more accessible. In the United States, the average yearly cost of orphan drugs is around $32,000, with more than one-third of drugs for rare conditions priced above $100,000 annually.
Europe is estimated to grow at the fastest rate in the market during the forecast period due to a focus on personalized medicine, increased investment in rare disease treatments, and cross-border healthcare initiatives that improve access. Public-private partnerships are accelerating research and drug approval, while more specialized treatment centers and advancements in health technology are enhancing accessibility. These factors are making Europe a key hub for orphan drug development and distribution. According to the European Medicines Agency (EMA), around 36 million people in the EU are affected by over 6,000 rare diseases, many of which impact fewer than one in 100,000 individuals. This growing prevalence of rare conditions, combined with Europe’s strong focus on supporting orphan drug development, has positioned the region as a key driver of growth in the orphan drug market.
The Germany Orphan Drug Market Trends
The German market is growing due to its robust healthcare system with wide insurance coverage, a supportive regulatory environment for drug approval and reimbursement, and a strong biotech sector driving innovation. Rising awareness of rare diseases, an increasing patient population, and Germany's strategic position in European healthcare further contribute to the expansion of the market.
The UK Orphan Drug Market Trends
The UK market is expanding due to a focus on personalized medicine and targeted therapies for rare diseases. The rise in precision healthcare, along with strong private-public partnerships and collaborations with research organizations, is driving orphan drug development. The increasing use of digital health technologies and telemedicine is improving patient access to treatments. Additionally, the UK's emphasis on rare disease awareness and patient-focused policies is fueling market growth as more patients and healthcare providers seek better treatment options.
Asia-Pacific is expected to see significant growth in the Orphan drug market during the forecast period due to rising healthcare investments, improved infrastructure, and increased awareness of rare diseases. Expanding regulatory frameworks, strategic partnerships in research, and growing demand for innovative therapies also contribute to the market's growth. As biotech industries expand, the region is becoming a key player in orphan drug development.
The China Orphan Drug Health Market
China’s market is growing due to increased healthcare investments, regulatory reforms that speed up drug approvals, and rising awareness of rare diseases. Government support, biotech innovation, and collaborations with global pharma companies are also driving market expansion. In China, at least 10 million people are affected by rare or orphan diseases, while globally, the number reaches around 350 million.
The India Orphan Drug Market Trends
The Indian market is expanding due to a growing awareness of rare diseases, improvements in healthcare infrastructure, and government support for orphan drug development. India’s large patient population, along with increasing biotech innovation and global pharmaceutical collaborations, is driving demand for treatments for rare conditions.
South America is expected to grow significantly in the orphan drug market during the forecast period, due to growing rare disease awareness, which is increasing their early diagnoses, driving the demand for orphan drugs. Growing government initiatives, pharmaceutical investments, and expanding healthcare are also increasing their use, enhancing the market growth.
Brazil Orphan Drug Market Trends
Brazil consists of stringent regulations that are increasing the orphan drug approvals, creating new opportunities for their innovations, where the growing health awareness and rare disease prevalence are also increasing their use. Additionally, increasing government support and industrial collaborations are also increasing their advancements.
MEA is expected to show lucrative growth in the orphan drug market during the forecast period, due to expanding healthcare infrastructure and regulatory reforms, which are increasing the adoption and accessibility of the orphan drugs. Moreover, rising reimbursement policies, R&D activities, and collaborations are also increasing their acceptance rates and innovations, which is promoting the market growth.
Saudi Arabia Orphan Drug Market Trends
Due to growing incidences of rare diseases across Saudi Arabia, the demand for orphan drugs is increasing, where the growing early diagnosis is also increasing their adoption rates. At the same time, expanding government policies, healthcare infrastructure, health awareness, and technological advancement are also increasing their use and innovations.
R&D
Clinical Trials and Regulatory Approvals
Packaging and Serialization
Distribution to Hospitals, Pharmacies
Patient Support and Services

By Drug Type
By Therapy
By Distribution Channel
By Region
April 2026
April 2026
April 2026
April 2026