April 2026

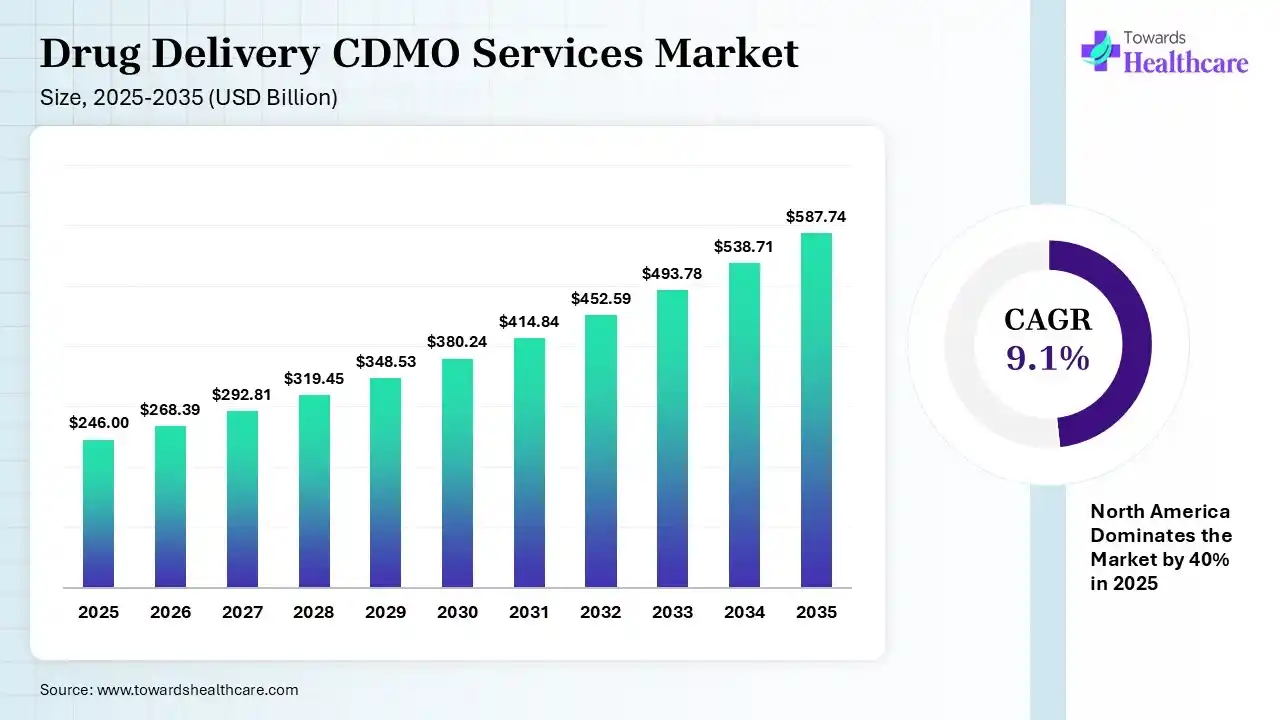
The global drug delivery CDMO services market size was estimated at USD 246 billion in 2025 and is predicted to increase from USD 268.39 billion in 2026 to approximately USD 587.74 billion by 2035, expanding at a CAGR of 9.1% from 2026 to 2035. Increasing demand for advanced treatment options is increasing the use of drug delivery CDMO services. Growing regulatory complexities, expanding drug delivery technologies, increasing collaborations, and new product launches are enhancing the market growth.
The drug delivery CDMO services market is driven by the increasing complexity of modern therapeutics and increasing outsourcing trends. The drug delivery CDMO services encompass the specialized services promoting the development, manufacturing, and optimization of the drug delivery system. The help in the development of advanced drug delivery systems, new dosage forms, complex formulations, and supports commercial manufacturing and clinical trials with regulatory compliance.
AI offers a wide range of applications in the drug delivery CDMO services market, promoting formulation and process optimization. It also offers drug release modelling and predictive analytics, which help in the development of stable and effective formulations. AI is also used for quality control, large-scale production, development of patient-specific formulations, and regulatory compliance, promoting faster and optimized production.
Growing Outsourcing Trends
A rise in the demand for integrated end-to-end CDMO services is increasing the outsourcing trends, promoting new collaborations. This is driving the outsourcing of various products to leverage their development, manufacturing, and packaging services.
Rise Of Advanced Therapies
The growing adoption of advanced treatment options like gene therapies, biologics, antibody drug conjugates, and mRNA therapy is increasing the innovations. This is promoting the use of drug delivery CDMO services. They offer new technologies like nanoparticles and lipid-based drug delivery systems, encouraging their adoption.
Technological Advancements
The growing technological innovations are driving the development of new drug development and manufacturing solutions. AI-powered formulation development, smart delivery devices, single-use systems, continuous manufacturing, on-body injectors, and modular facilities are being developed.
| Table | Scope |
| Market Size in 2026 | USD 268.39 Billion |
| Projected Market Size in 2035 | USD 587.74 Billion |
| CAGR (2026 - 2035) | 9.1% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Drug Type, By End-user, By Technology, By Region |
| Top Key Players | Lonza, Vetter Pharma, Thermo Fisher Scientific, Kindeva, Catalent, Lubrizol Life Science, Recipharm, West Pharma, Samsung Biologics, Evonik |
| Segment | Share 2025 (%) |
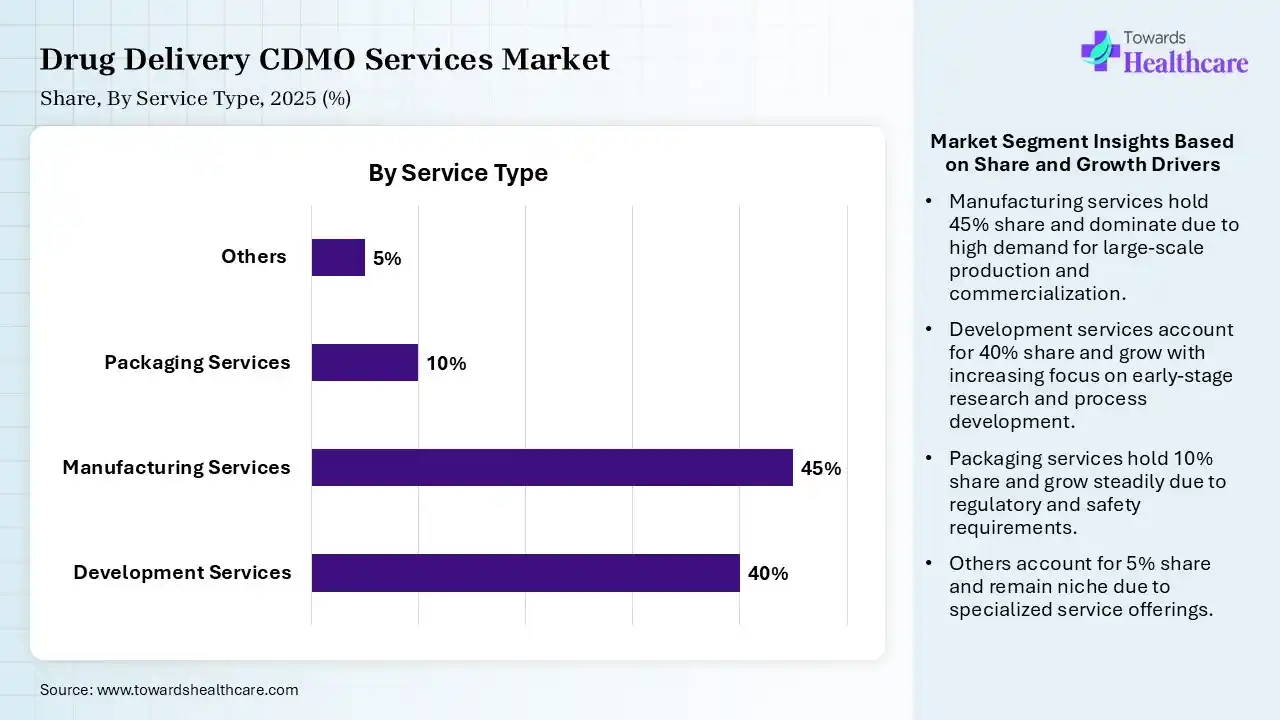
| Development Services | 40% |
| Manufacturing Services | 45% |
| Packaging Services | 10% |
| Others | 5% |
The Manufacturing Services Segment Dominated the Market With 45% in 2025
The manufacturing services segment led the drug delivery CDMO services market with 45% share in 2025, due to growth in the demand for commercial-scale manufacturing. A rise in the demand for affordable large-scale production also increased their use. The growth in the demand for generics and biologics has also increased their use.
The development services segment led the market with 40% share in 2025, and is expected to achieve the highest CAGR of 10% during the forecast period due to high large-scale production rates. The growth in the approval rates also increased their demand for commercial-scale manufacturing. Their established infrastructure, regulatory expertise, and advanced technologies are also increasing their use for the manufacturing of complex formulations.
The packaging services segment held 10% of the drug delivery CDMO services market share in 2025, driven by growing regulatory pressure on packaging standards. Growing temperature-sensitive products are also increasing the demand for these services. Growing technological innovations are also increasing their advantages.
The others (e.g., regulatory affairs) segment is expected to grow significantly in the market share in 2025, due to growing regulatory complexities. The increasing development of new drug products is also increasing their use for faster approvals. Frequent guideline updates also promote their use.
| Segment | Share 2025 (%) |
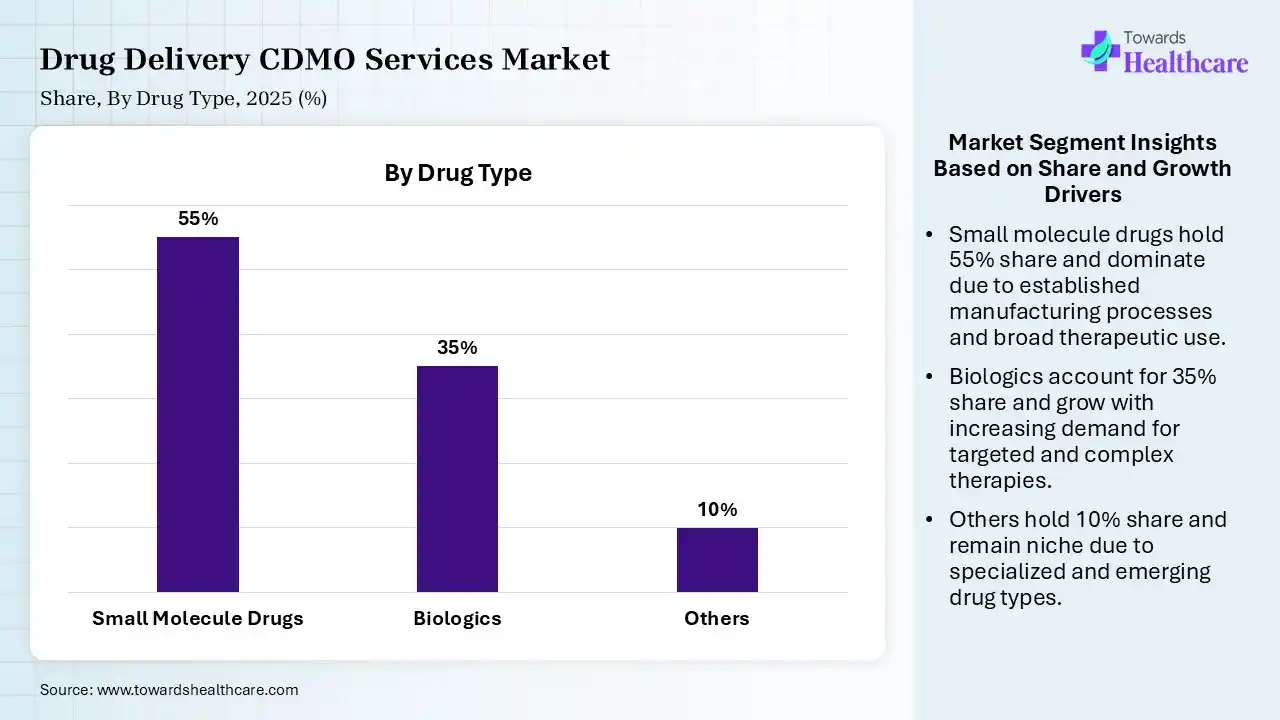
| Small Molecule Drugs | 55% |
| Biologics | 35% |
| Others | 10% |
The Small Molecule Drugs Segment Dominated the Market With 55% in 2025
The small molecule drugs segment accounted for the highest revenue share of 55% of the drug delivery CDMO services market in 2025, due to the presence of large volumes of small molecule therapies and treatment options. Their cost-effective manufacturing and development also increased the use of drug delivery CDMO services. The growth in the demand for generic products also increased their use.
The biologics segment held the second-largest share of 35% of market share in 2025, due to rapid advancements in biotechnology and gene therapy. Growing chronic disease burden and patent expiration are also increasing the use of drug delivery CDMO services. Their advanced technologies and expertise are also increasing their adoption.
The gene and cell therapy products sub-segment held 10% of the market share in 2025 and is expected to show the highest CAGR growth during the forecast period, due to a surge in cell-based therapies for rare diseases. Increasing incidences of cancer and rare diseases are also increasing their demand. This is driving the use of CDMO services for the development of new therapies.
The others (e.g., biosimilars) segment held 10% of the drug delivery CDMO services market share in 2025, due to growing focus on developing new biosimilars. Growing government support is also encouraging the use of drug delivery CDMO services. Increasing approvals and demand for cancer therapeutics are also increasing their use.
| Segment | Share 2025 (%) |
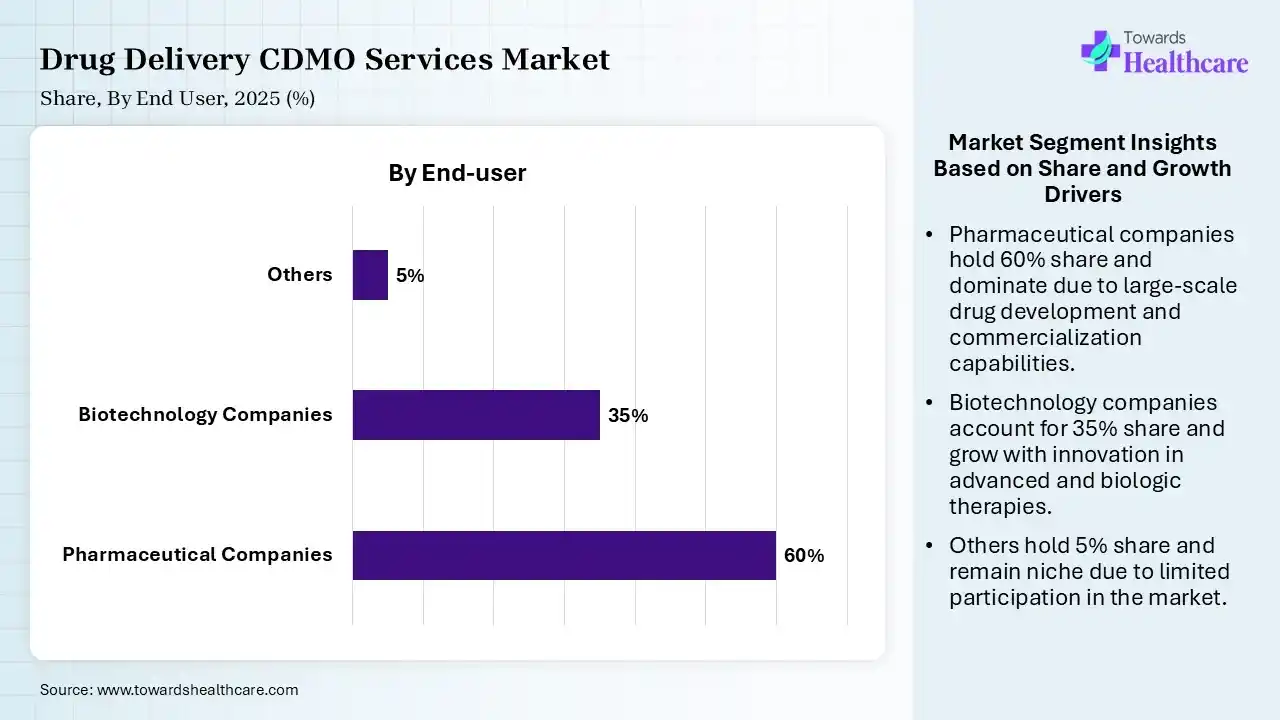
| Pharmaceutical Companies | 60% |
| Biotechnology Companies | 35% |
| Others | 5% |
The Pharmaceutical Companies Segment Dominated the Market With 60% in 2025
The pharmaceutical companies segment contributed the biggest revenue share of 60% of the drug delivery CDMO services market in 2025, due to the extensive use of CDMO for drug development and production. The presence of complex drug portfolios also increased their use. The growth in the development of new drugs and biologics has also increased their use.
The biotechnology companies segment held the second-largest share of 35% of the market in 2025, due to the rising number of biotech companies using contract manufacturing. Increasing demand for biologics and oncology treatments is also increasing the collaboration with CDMOs. Rising precision medicine demand is also increasing its use.
The emerging biotech sub-segment held 20% of the market share in 2025 and is expected to expand with the highest CAGR during the forecast period, due to increasing funding and research for novel therapies. Growing chronic disease burden and high unmet needs are also increasing the development of new products, driving the demand for CDMO services. Their advanced technologies are also attracting companies.
The others (e.g., contract research organizations) segment held 5% of the drug delivery CDMO services market share in 2025, driven by their rapid expansion. Increasing outsourcing trends are also promoting their use. Expanding clinical trials are also increasing the use of drug delivery CDMO services.
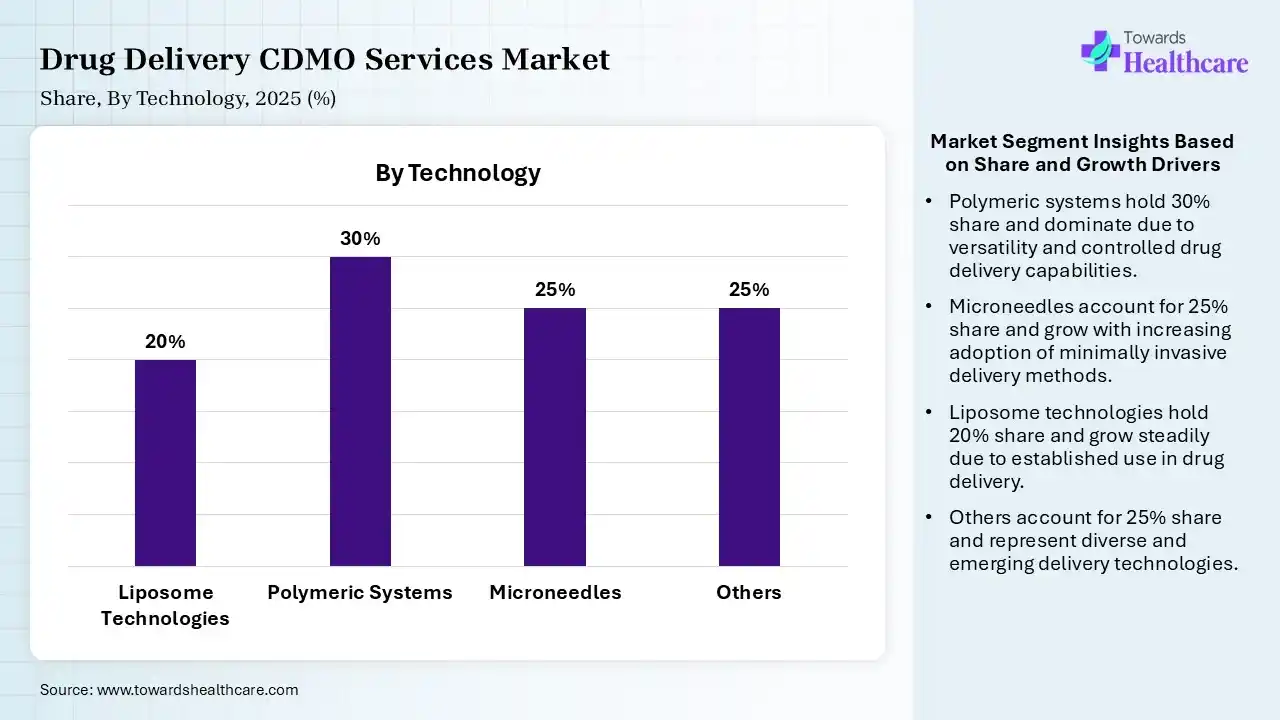
| Segment | Share 2025 (%) |
| Liposome Technologies | 20% |
| Polymeric Systems | 30% |
| Microneedles | 25% |
| Others | 25% |
The Polymeric Systems Segment Dominated the Market With 30% in 2025
The polymeric systems segment held the largest revenue share of 30% of the drug delivery CDMO services market in 2025, due to high demand for drug release-controlled delivery. Their high compatibility and improved stability also increased their use, which promoted the adoption of drug delivery CDMO services. Their affordable and targeted delivery capabilities also increased their use.
The microneedles segment held the second-largest share of 25% of the market in 2025 and is expected to grow with the fastest CAGR during the forecast period, due to growing demand for painless and efficient drug delivery. Increasing shift towards minimally invasive delivery is also increasing their use. Additionally, growing self-administration trends are also driving their demand.
The liposome technologies segment held 20% of the market share in 2025, due to its use in improving the bioavailability of drugs. They also help in enhancing the biocompatibility and stability of the formulations. Their growing applications in oncology and vaccine development are also increasing their demand.
The others (e.g., transdermal patches) segment held 25% of the drug delivery CDMO services market share in 2025, due to growing demand for non-invasive delivery. Their fewer side effects and sustained release profile are also increasing their use. This, in turn, is driving the adoption of drug delivery CDMO services.
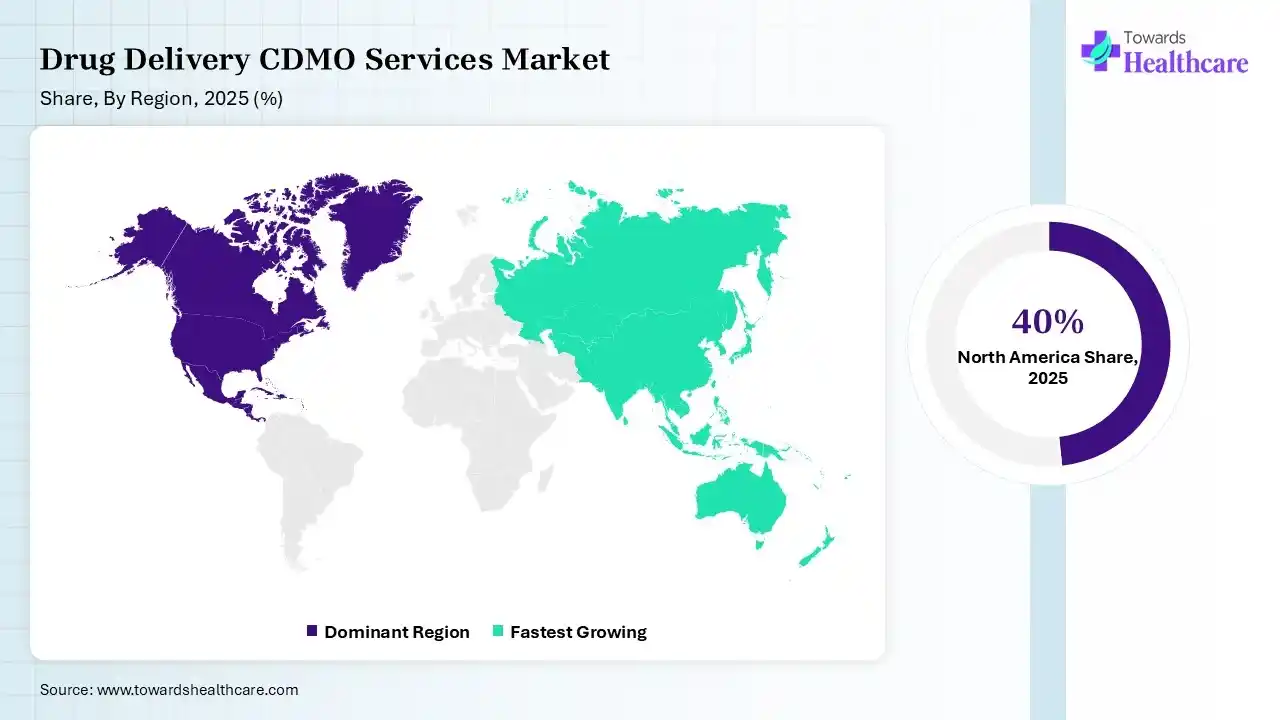
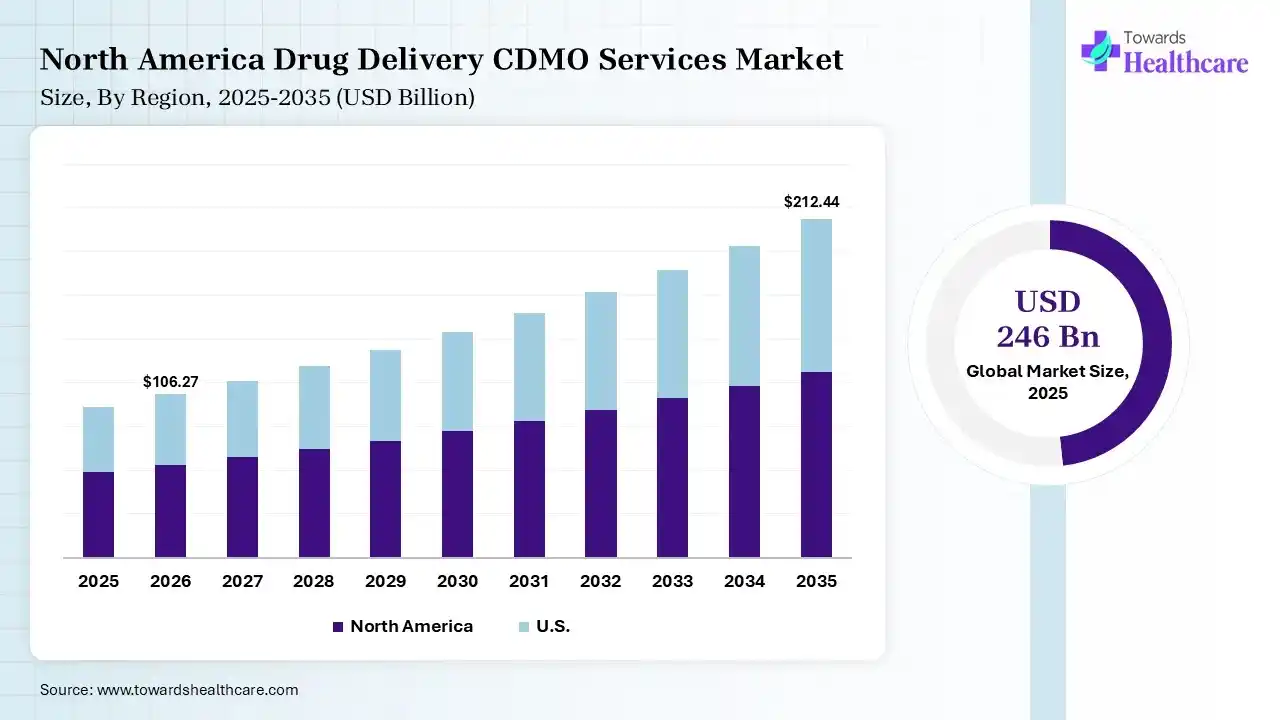
North America dominated the drug delivery CDMO services market with 40% in 2025, due to strong regulatory support for drug development and manufacturing. The presence of robust pharmaceutical industries and R&D infrastructure also increased the use of drug delivery CDMO services. The growth in outsourcing trends also contributed to the market growth.
U.S. Market Trends
A rise in pharmaceutical outsourcing across the U.S. is increasing the use of drug delivery CDMO services. The presence of robust industries and high R&D investments is also promoting innovations, driving their demand. The increasing demand for advanced therapies and drug delivery technologies is also increasing their use.
Asia Pacific held 20% share of the drug delivery CDMO services market in 2025 and is expected to grow at the fastest CAGR during the forecast period, due to rapid expansion in pharmaceutical manufacturing. The growing chronic disease burden is also increasing the use of drug delivery CDMO services for new drug formulations. Expanding clinical trials are also enhancing the market growth.
India Market Trends
The expanding pharmaceutical and CDMO infrastructure in India is increasing the use of drug delivery CDMO services. Expanding generic drug manufacturing and outsourcing trends are also increasing their demand. Their affordable services and expertise are also promoting their adoption rates.
R&D
Packaging and Serialization
Patient Support and Services

| Companies | Headquarters | Drug Delivery CDMO Services |
| Lonza | Basel, Switzerland | Parenteral Drug Product Services |
| Vetter Pharma | Ravensburg, Germany | Aseptic pre-filled syringes, cartridges, and dual-chamber bypass systems |
| Thermo Fisher Scientific | Waltham, U.S. | Patheon Services, Patheon Logistics, and Quick to Clinic |
| Kindeva | Woodburg, U.S. | Microneedle patches, metered-dose inhalers, and transdermal delivery |
| Catalent | Somerset, U.S. | OneBio and GPEx Boost |
| Lubrizol Life Science | Cleveland, U.S. | Ophthalmic/topical formulation and Apisolex |
| Recipharm | Stockholm, Sweden | ReciBioPharm and Recipharm Advanced Therapy |
| West Pharma | Exton, U.S. | SmartDose and Self-injection Systems |
| Samsung Biologics | Incheon, South Korea | mRNA Production and Live-Cell Imaging |
| Evonik | Essen, Germany | Eudragit polymer and bioresorbable implants |
Strengths
Weaknesses
Opportunities
Threats
By Service Type
By Drug Type
By End-user
By Technology
By Region
April 2026
April 2026
April 2026
April 2026