April 2026

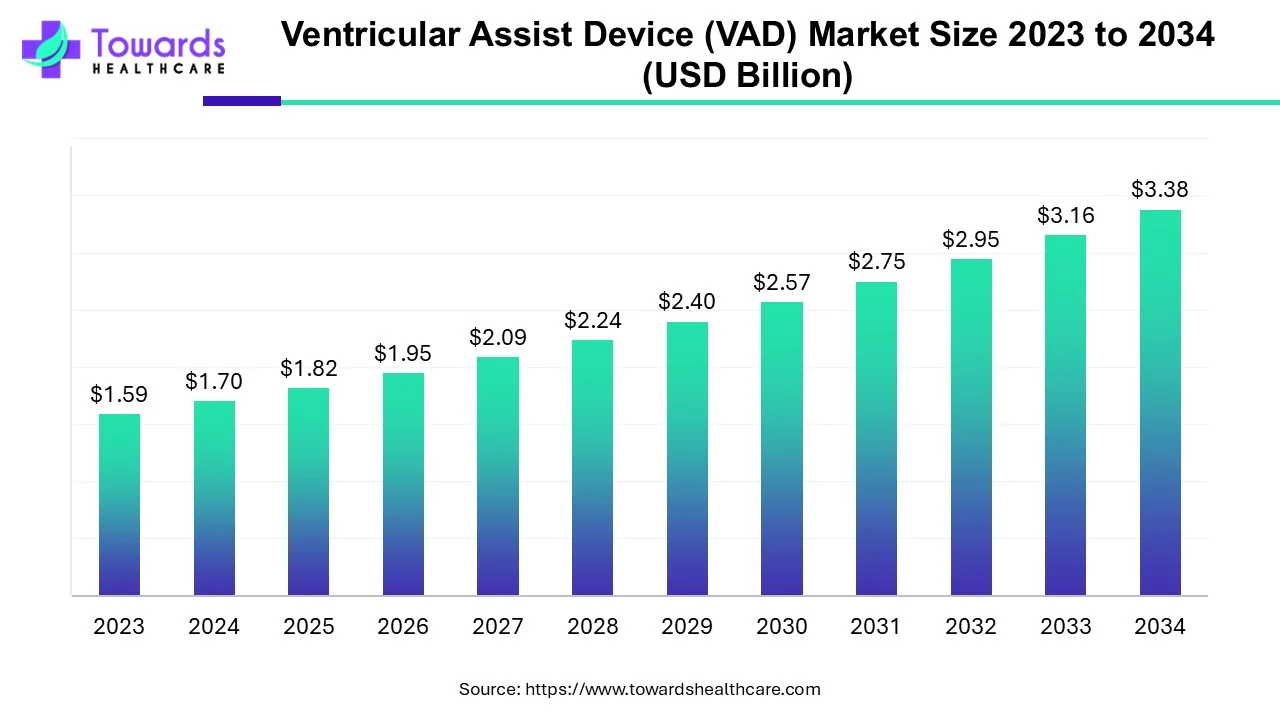
The Ventricular Assist Device (VAD) market size is projected to grow from USD 1.82 billion in 2025 to USD 3.38 billion by 2034, at a compound annual growth rate (CAGR) of 7.1%.
A ventricular assist device (VAD) is a device that helps in pumping healthy blood from lower chambers (ventricles) in the heart to all parts of the body. VAD is used when someone’s heart is unable to pump a sufficient amount of blood or healthy blood, and it is inserted by surgery. VAD can give life to patients whose hearts are too weak to perform their normal functions. VAD is a tiny implant, after implantation of, patients experience betterment in the symptoms of their heart function and improved quality of life which can last for several years. LVAD which is the left ventricular assist device is the most commonly used and preferred VAD which pumps oxygen-rich blood from the left ventricle to all parts of the body.
In addition, RVAD which is the right ventricular assist device is another device implanted to pump oxygen-poor blood from the right ventricle in the heart to the lungs for oxygenation. Furthermore, biventricular assist devices and pediatric VADs are other devices used in the surgery. VAD can help patients temporarily who are going to undergo heart transplant surgery and waiting for a heart from a donor to be available. On the other hand, VAD may be used permanently to assist the heart in pumping blood. Moreover, this procedure can be helpful for patients who cannot undergo heart transplant surgery because of age and other factors. This explains the destination therapy. Destination therapy can help patients live longer with heart failure and have age-related or other risk factors. Besides this, in the bridge to recovery therapy LVAD is recommended by doctors to help patients with temporary heart failure until their heart comes back to normal pumping.
Durable left ventricular assist devices (LVADs) have transformed the management of progressive heart failure, meaningfully enhancing the quality of life and survival rate. Over the years, LVADs have developed from first-generation pulsatile tools to advanced wholly magnetically levitated (mag-lev) pumps. Durable implantable left ventricular assist devices (LVADs) have been shown to improve the quality and survival of life for patients with stage D cardiac failure, which has caused the growth of the ventricular assist device market.
The global VAD (ventricular assist device) surgery treatment market is expected to develop significantly, with rising heart failure rates serving as a primary driver. As the world's population ages and life expectancy rises, so does the number of people at risk for heart failure. This chronic illness impairs the heart's capacity to pump blood efficiently, resulting in exhaustion, shortness of breath, and other severe symptoms.
Patients with severe heart failure might have exhausted traditional therapy options, such as drugs and lifestyle changes. In such circumstances, VAD surgery can save a person's life. VADs serve as mechanical pumps implanted in the chest, helping or replacing the heart's pumping function while dramatically boosting blood flow throughout the body.
VADs help patients awaiting heart transplants by keeping them alive while they wait for a donor organ. They can also be used as destination therapy for patients who are not candidates for transplants, extending their lives and enhancing their quality of life.
VADs relieve symptoms such as weariness and shortness of breath by providing proper blood circulation, allowing patients to resume their regular activities with renewed vitality. This allows people to participate more completely in life and significantly improves their well-being.
The rising incidence of heart failure results in a larger pool of potential patients who could benefit from VAD therapy. This, in turn, drives up demand for VAD devices, surgical operations, and other healthcare services. As more people seek this treatment option, the vascular assist device market continues to increase.
The increasing burden of heart failure globally presents a significant challenge, but it also highlights the crucial role VAD technology plays in improving patient outcomes. With continued advancements in VAD design, surgical techniques, and access to care, the vascular assist device market is well-positioned for sustained growth in the years to come.
The vascular assist device market is expanding due to increased demand for destination therapy. Historically, VADs were largely utilized as bridge-to-transplant devices, keeping patients alive while they waited for a suitable heart donor. However, the paucity of donor organs has resulted in a paradigm change.
Destination therapy uses VADs as a long-term option for patients with severe heart failure who are not candidates for transplantation. By providing a viable alternative to transplants, VADs reduce the strain on the donor pool, potentially saving more lives. VADs dramatically improve patient survival rates and quality of life as compared to advanced heart failure therapy with medicines alone. Destination therapy broadens the patient pool eligible for VAD implantation, providing hope to a broader variety of people with severe heart failure.
The increased demand for destination therapy translates directly into market growth. As more patients choose long-term VAD therapy, the demand for VAD devices, surgical procedures, and related healthcare services grows. This tendency is projected to continue, driving the vascular assist device market market ahead.
The vascular assist device market industry is being pushed not just by the need for the technology, but also by advances in surgical procedures. A crucial motivator in recent years has been the emphasis on minimally invasive VAD implantation methods.
Traditional VAD implantation required open-heart surgery, which resulted in lengthier hospital stays and extended recovery times. Minimally invasive procedures, such as mini-thoracotomy or robotic-assisted surgery, necessitate smaller incisions and cause less disruption in the chest cavity. This results in speedier healing times, less post-surgical pain, and a quicker return to normal activities for patients.
Minimally invasive techniques are less physically taxing on patients, resulting in less discomfort and anguish than traditional open-heart surgery. This can dramatically improve the entire patient experience and encourage more people to think about VAD therapy as a potential treatment option.
The advantages of speedier recovery and less discomfort are driving an increase in VAD surgery acceptance rates. As minimally invasive treatments become more standardized and generally available, more patients and surgeons are likely to adopt VAD therapy, increasing market reach. The emphasis on less invasive VAD implantation represents a considerable opportunity for market expansion. With speedier procedures, shorter hospital stays, and better patient outcomes, the Vascular assist device market market becomes more appealing to both patients and healthcare providers. This tendency is projected to continue, accelerating market growth in the following years. While minimally invasive procedures have many advantages, they may not be appropriate for all patients. Surgeon expertise and patient-specific considerations will continue to influence the most suited surgical strategy.
The vascular assist device market has enormous potential for improving the lives of people with severe heart failure. However, the expensive expense of the entire treatment process poses a considerable barrier to its widespread adoption. This monetary burden affects both people and healthcare systems.
The ventricular assist device is a complicated piece of medical equipment. The initial price of these gadgets is considerable due to research, development and manufacturing costs. Implanting a VAD necessitates specialized surgical skills and operating room resources, resulting in hefty surgical expenditures.
The high expense of VAD therapy puts pressure on healthcare finances, potentially reducing resources available for other critical medical treatments.
While Western markets now dominate the vascular assist device market landscape, developing countries in Asia-Pacific, Latin America, and the Middle East offer considerable opportunities in their rising healthcare sectors. Heart failure is projected to become more common as these regions' populations develop and their demographics age. This creates a larger pool of possible patients who may benefit from VAD therapy. Developing countries frequently lack established VAD programs, allowing market participants to emerge and establish themselves as significant suppliers. This can include forming alliances with local hospitals and healthcare organizations to promote access to VAD technology and expertise. The development of new VADs with longer lifespans, reduced production costs, and maybe easier surgical procedures may make them more appealing to resource-constrained healthcare systems in these locations.
The vascular assist device market market's growth and adoption rates vary by geographical region. Developed countries have a higher prevalence of heart failure due to an aging population and lifestyle choices. This corresponds to a larger pool of possible VAD patients and a more mature VAD surgery market.
Developed countries frequently have more comprehensive healthcare coverage and favorable reimbursement rules for VAD therapy, which increases patient access. These regions have well-equipped facilities, trained surgeons, and established VAD programs, resulting in greater adoption rates. The developed markets are at the forefront of VAD research and development, with an emphasis on developing more advanced and cost-effective devices.
Developing Asia Pacific countries are seeing an increase in heart failure cases, generating a potential market for VAD therapy. These regions may have limited access to specialized surgeons, VAD devices, and advanced medical facilities, stifling market expansion. VAD therapy reimbursement rules may be less developed or restrictive in developing nations, creating a financial hurdle for patients. Healthcare resources are generally tighter, therefore there is a higher emphasis on cost-effective solutions, which may favor less expensive VAD options if available.

(**We also provide cross-sectional analysis in market segments)
By Device Type
By Application
By End User
By Geography
April 2026
April 2026
April 2026
April 2026