May 2026

The global hearing loss gene therapy market size was estimated at USD 1.12 billion in 2025 and is predicted to increase from USD 1.33 billion in 2026 to approximately USD 6.32 billion by 2035, expanding at a CAGR of 18.9% from 2026 to 2035. A huge rise in the geriatric population, who are highly susceptible to hearing loss conditions, & rising funding in R&D and clinical trials, drive the overall market development. Eventually, the market will adopt gene editing approaches, like CRISPR Cas9, which fosters innovations in the hearing loss issues.
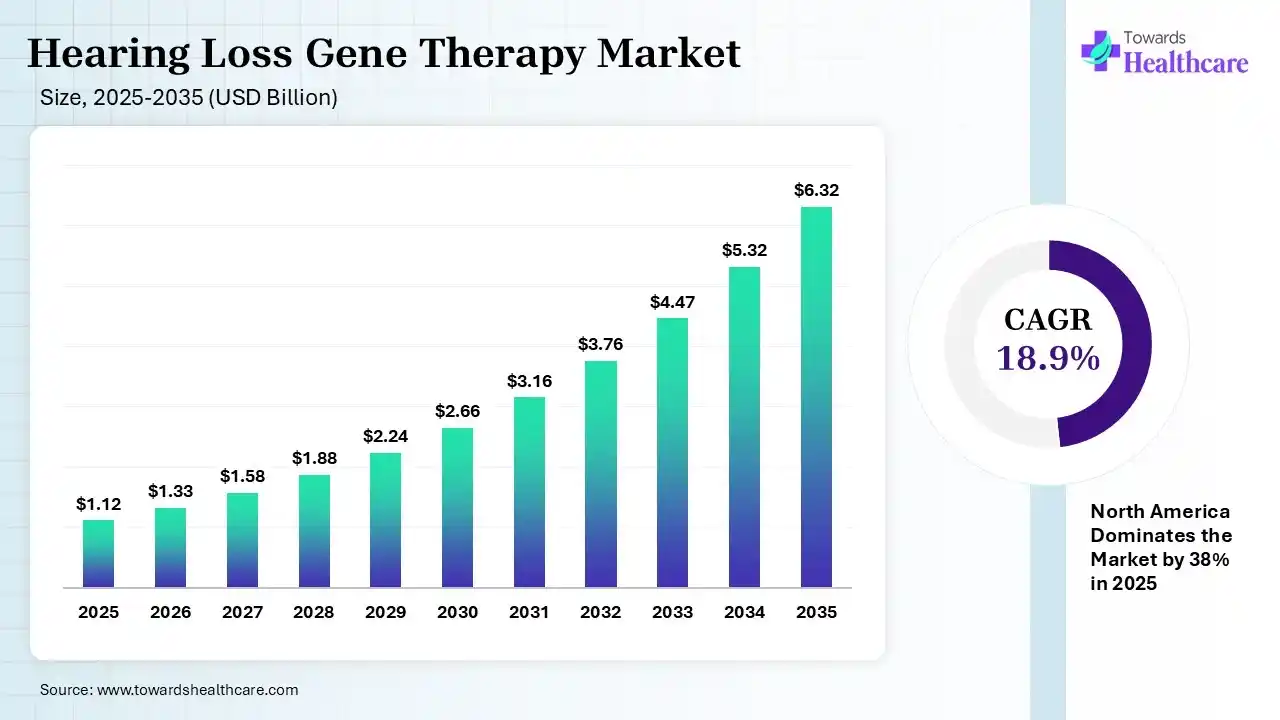
Primarily, the hearing loss gene therapy market is defined as the emergence of novel approved treatments to treat genetic issues that lead to deafness by employing functional genes into the inner ear. Whereas the global market progression is propelled by the massive rise in congenital, noise-induced, & age-related impairment, with increasing clinical trials success & FDA authorizations. Alongside, the development of targeted gene therapies to reach the main cause of inherited deafness is impacting the overall demand.
Specifically, the use of machine learning algorithms assists in estimating hereditary hearing loss from specific gene variants, including GJB2 & SLC26A4. Besides this, AI solutions support designing adeno-associated viruses (AAVs) for the efficient delivery of functional genes into inner ear hair cells. However, emerging new AI tools are facilitating deeper, 3D views of hair cell stereocilia, enabling researchers to assess how effectively gene therapy rescues damaged auditory structures.
Immersive Clinical Advances
Particularly, the market is emphasizing treatment for OTOF-related congenital deafness, while the FDA has approved Otarmeni, which enables a dual AAV vector to deliver a functional OTOF gene, resulting in a major enhancement in speech & hearing.
Fostering Vector Technology & Delivery
Nowadays, researchers are preferring recombinant Adeno-associated virus (rAAV) due to its safety, efficiency, & ability to be delivered directly into the inner ear via the round window membrane.
Spurring Non-Viral Vector Progression
Eventual studies are seeking into non-viral alternatives, such as nanoparticles or cationic lipids that enable repeated dosing.
| Table | Scope |
| Market Size in 2026 | USD 1.33 Billion |
| Projected Market Size in 2035 | USD 6.32 Billion |
| CAGR (2026 - 2035) | 18.9% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Therapy Type, By Vector Type, By Hearing Loss Type, By Delivery Method, By End User, By Region |
| Top Key Players | Regeneron Pharmaceuticals, Akouos (Eli Lilly), Sensorion, Fudan University, Otovia Therapeutics, Astellas Pharma, BridgeBio Pharma |
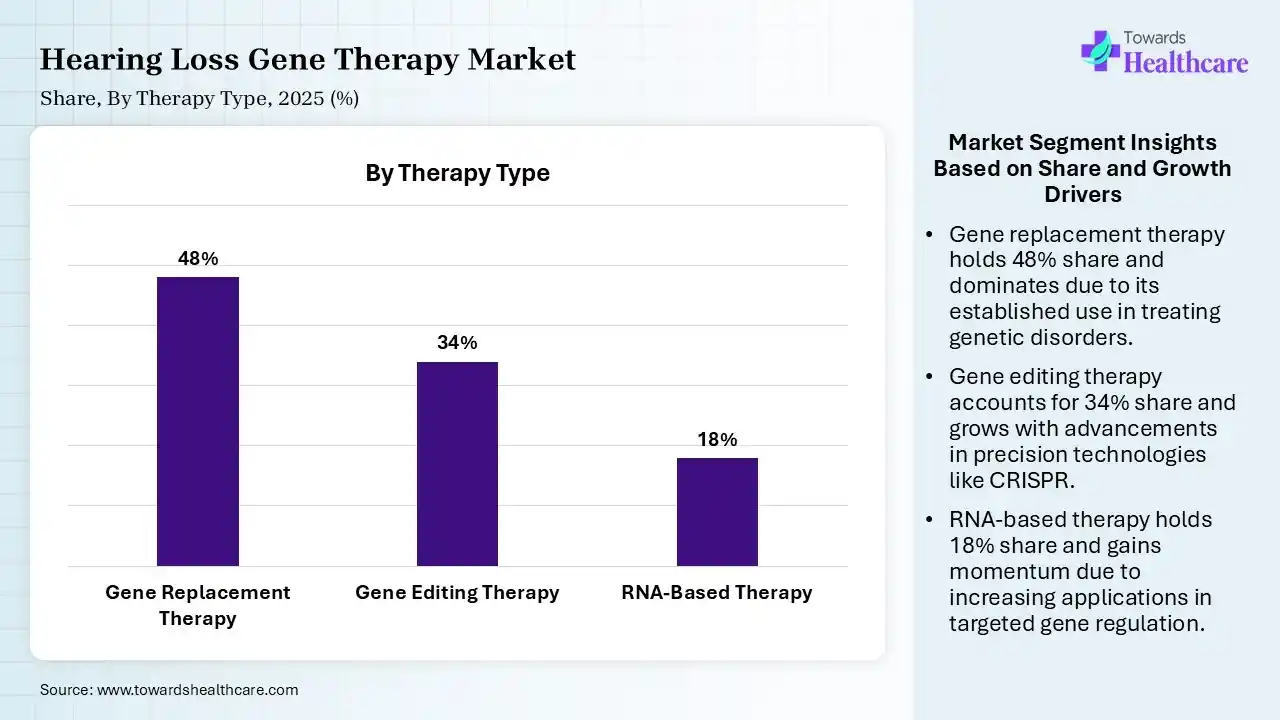
| Segment | Share 2025 (%) |
| Gene Replacement Therapy | 48% |
| Gene Editing Therapy | 34% |
| RNA-Based Therapy | 18% |
The Gene Replacement Therapy Segment Dominated the Market in 2025
In 2025, the gene replacement therapy segment held a dominant share of 48% of the hearing loss gene therapy market. Prominent catalysts include successful pre-clinical restoration of hearing in animal models and the ongoing evolution of safe AAV vectors for inner ear delivery. Whereas biological restoration offers extensive frequency resolution & pitch discrimination.
Moreover, the gene editing therapy segment captured the second largest share of a 34% in 2025 & is predicted to expand fastest. Globally surging cases of monogenic causes, breakthroughs in CRISPR/Cas9 technologies, & the inner ear’s accessibility for local, targeted delivery, fuel the demand for this therapy type. The latest study has shown the efficiency of CRISPR-based editing to treat DFNA41, which restores both hearing & balance in preclinical models.
The RNA-based therapy segment held 18% share of the hearing loss gene therapy market, as they facilitate reversible modulation of gene expression. Moreover, expanded research activities in antisense technologies & strengthened clinical trials are bolstering the therapeutic purposes of this type.
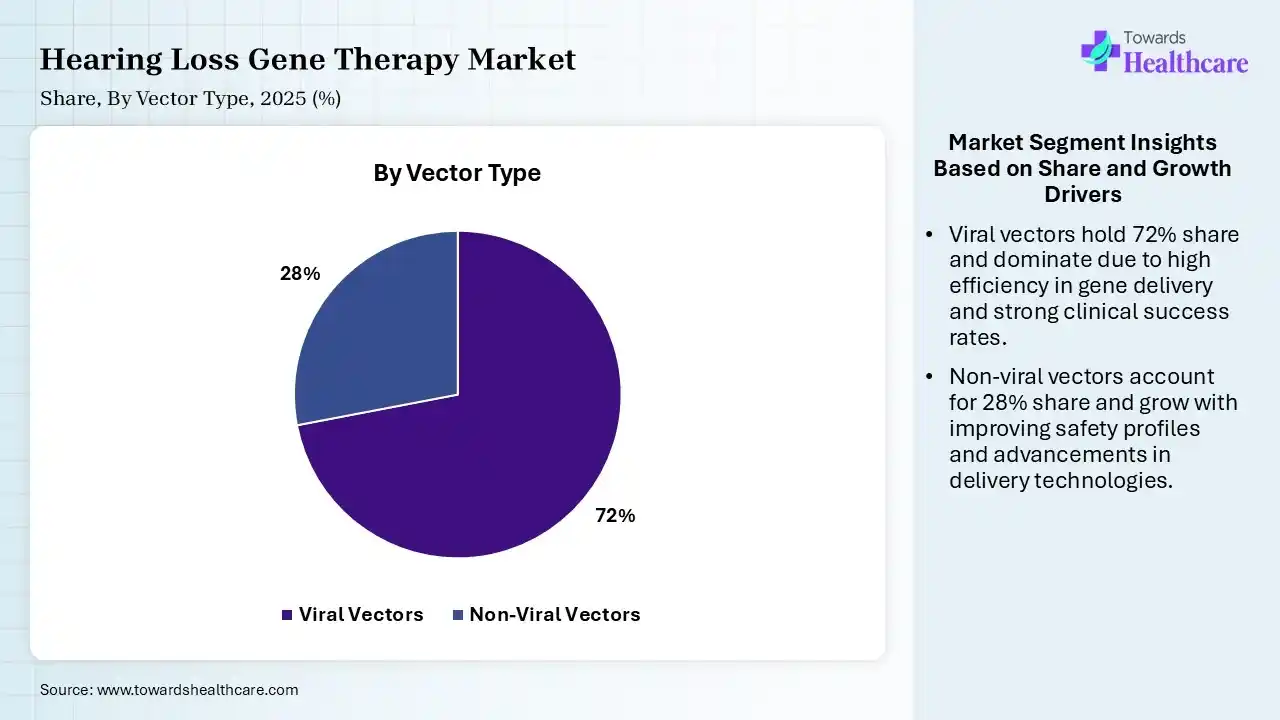
| Segment | Share 2025 (%) |
| Viral Vectors | 72% |
| Non-Viral Vectors | 28% |
The Viral Vectors Segment Led the Market in 2025
The viral vectors segment captured the largest share of a 72% of the market in 2025. Its dominance is propelled by its low immunogenicity, safety, & ability to transduce both dividing & non-dividing cells. Alongside, these vectors have a pivotal role in treating genetic sensorineural hearing loss, with Anc80L65, AAV1, & AAV2/9 being significant serotypes for targeting hair cells & assisting cells.
On the other hand, the non-viral vectors segment is anticipated to witness rapid expansion in the hearing loss gene therapy market. They have a high safety profile, with specialized carriers, such as lipid nanoparticles (LNPs) & polymeric carriers. The market is shifting towards breakthroughs in silica & gold nanoparticles, enabling non-invasive, targeted delivery of genetic materials.
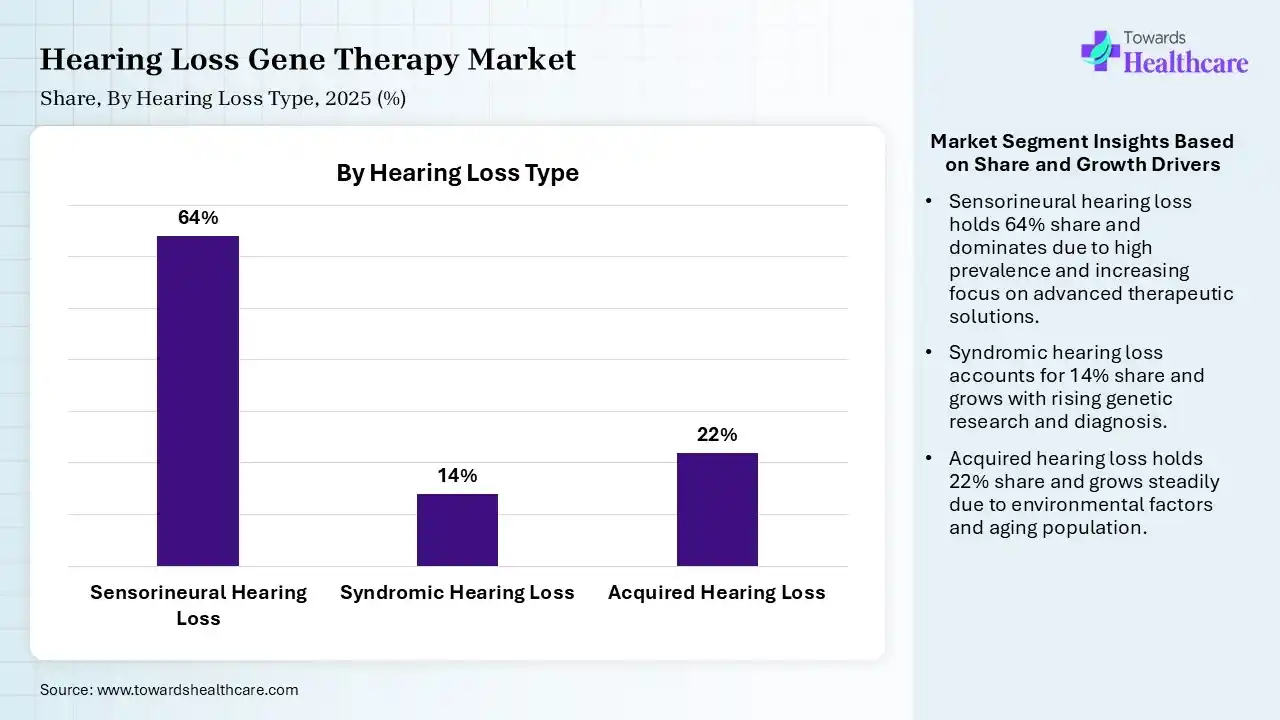
| Segment | Share 2025 (%) |
| Sensorineural Hearing Loss | 64% |
| Syndromic Hearing Loss | 14% |
| Acquired Hearing Loss | 22% |
The Sensorineural Hearing Loss Segment Was Dominant in the Market in 2025
In 2025, the sensorineural hearing loss segment led with a 64% share of the market. This kind is a permanent one, which also develops from genetic factors, head trauma, tumors, or autoimmune diseases. Researchers are investigating novel targeted therapies for various kinds of hereditary deafness, like Usher syndrome type 1C (USH1C) & connexin 26 (GJB2).
The acquired hearing loss segment held a 22% share in 2025 & is estimated to expand rapidly in the hearing loss gene therapy market. This type is fueled by higher exposure to sound greater than 85 dB, with side effects of ototoxic drugs, & viral infections/issues, like meningitis, mumps, & measles, can cause damage, especially in children.
In 2025, the syndromic hearing loss segment accounted for 14% share, due to the notable genetic mutations, such as autosomal dominant, recessive, or mitochondrial, connected with abnormalities in other body systems. Researchers are highly focused on Usher Syndrome by employing AAV vectors to deliver healthy genetic material to the inner ear.
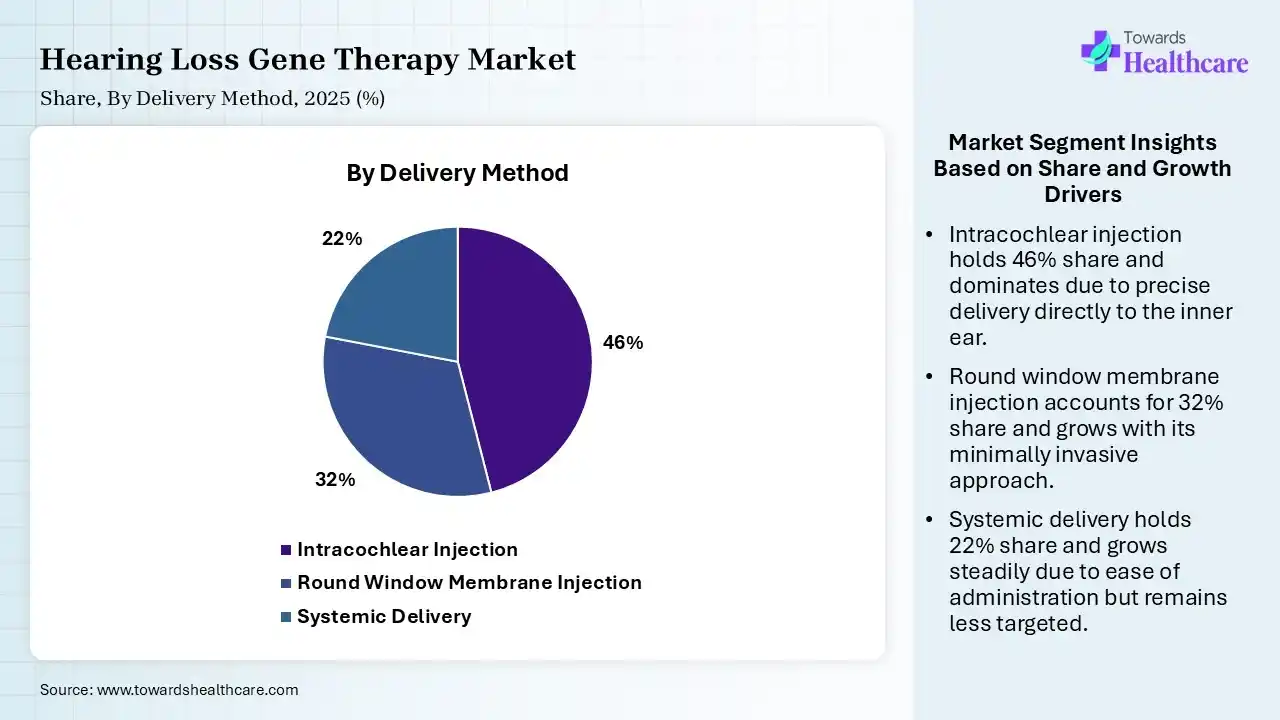
| Segment | Share 2025 (%) |
| Intracochlear Injection | 46% |
| Round Window Membrane Injection | 32% |
| Systemic Delivery | 22% |
The Intracochlear Injection Segment Dominated the Market in 2025
The intracochlear injection segment captured a 46% share of the hearing loss gene therapy market in 2025. Their dominance is driven by a direct, localised approach to restoring biological hearing & also targets the cellular roots of deafness. The leading organizations are using this method to treat hereditary sensorineural hearing loss (SNHL), which delivers viral vectors directly into the fluid-filled cochlea.
The round window membrane injection segment accounted for 32% share in 2025 & is anticipated to show the fastest growth. This is a minimally invasive approach, which improves patient safety, coupled with the emergence of delivery techniques, drives the overall adoption. This method is often unified with posterior semicircular canal fenestration (RWM+CF) to boost transduction in adult models, & enable wider expression.
However, the systemic delivery segment held a 22% share of the hearing loss gene therapy market. Respective expansion is fueled by its offerings, including a non-invasive option for gene therapy, with advances in targeting mechanisms to enhance specificity.
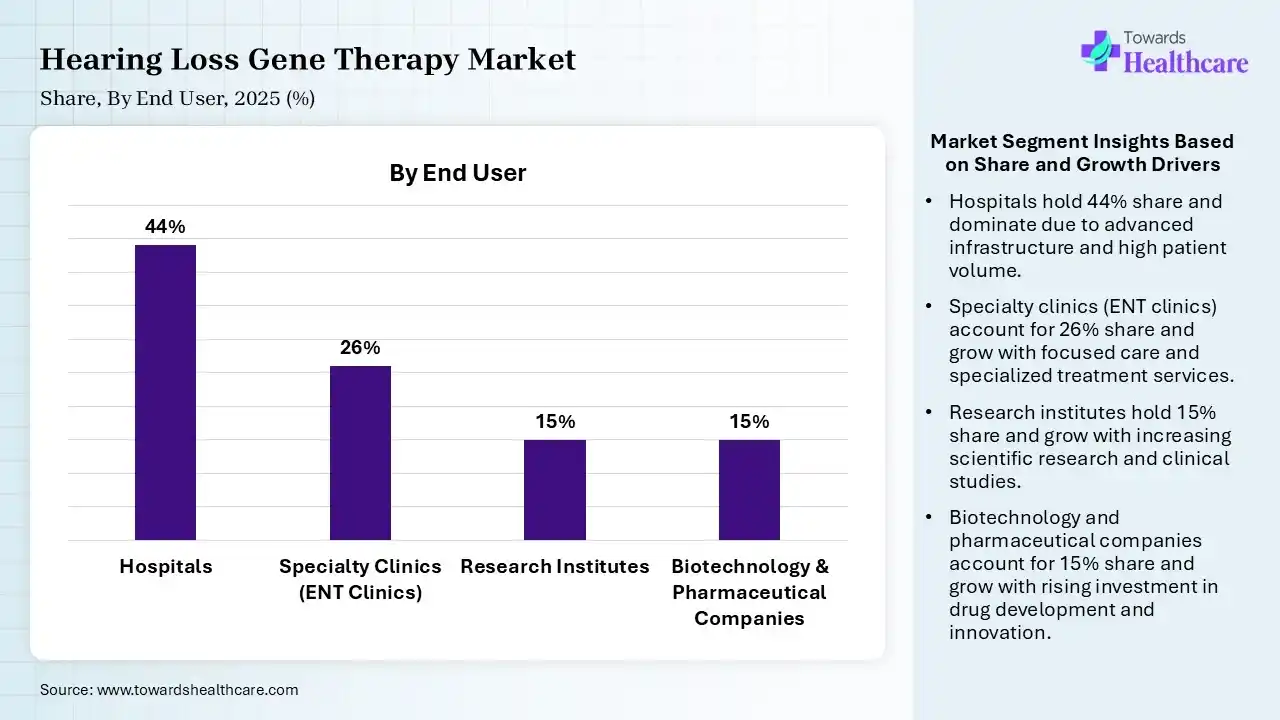
| Segment | Share 2025 (%) |
| Hospitals | 44% |
| Specialty Clinics (ENT Clinics) | 26% |
| Research Institutes | 15% |
| Biotechnology & Pharmaceutical Companies | 15% |
The Hospitals Segment Led the Market in 2025
In 2025, the hospitals segment dominated with a 44% share of the market. Primarily, hospitals provide rigorous ENT specialists, audiologists, & more sophisticated diagnostics. Besides this, hospitals offer robust diagnosis, cochlear implant surgery, hearing aid fitting, & rehabilitative therapy to boost quality of life & communication. They widely facilitate surgical, in vivo, round-window membrane injections.
The specialty clinics (ENT clinics) segment held the second-largest share of 26% of the hearing loss gene therapy market in 2025. These clinics are increasingly offering specialized, multi-disciplinary, & highly tailored care necessary for delicate inner ear treatments. They have a key role in finding genetic causes, accurate surgical delivery of therapeutics, & long-term monitoring.
However, the research institutes segment held 15% of the market share and is estimated to grow fastest in the market. Their expansion is promoting the transformation of viral vectors for precise gene delivery, performing vital preclinical trials on animal models, & exploring clinical trials to test safety & effectiveness. Many research institutes are working on CRISPR Cas9, gene editing approaches, optogenetics & gene therapy.
The biotechnology & pharmaceutical companies segment held a 15% share in 2025, due to the robust investment in R&D. Also, these companies are bolstering strategic alliances to raise commercialization, and emphasizing the progression of the biotech hub.
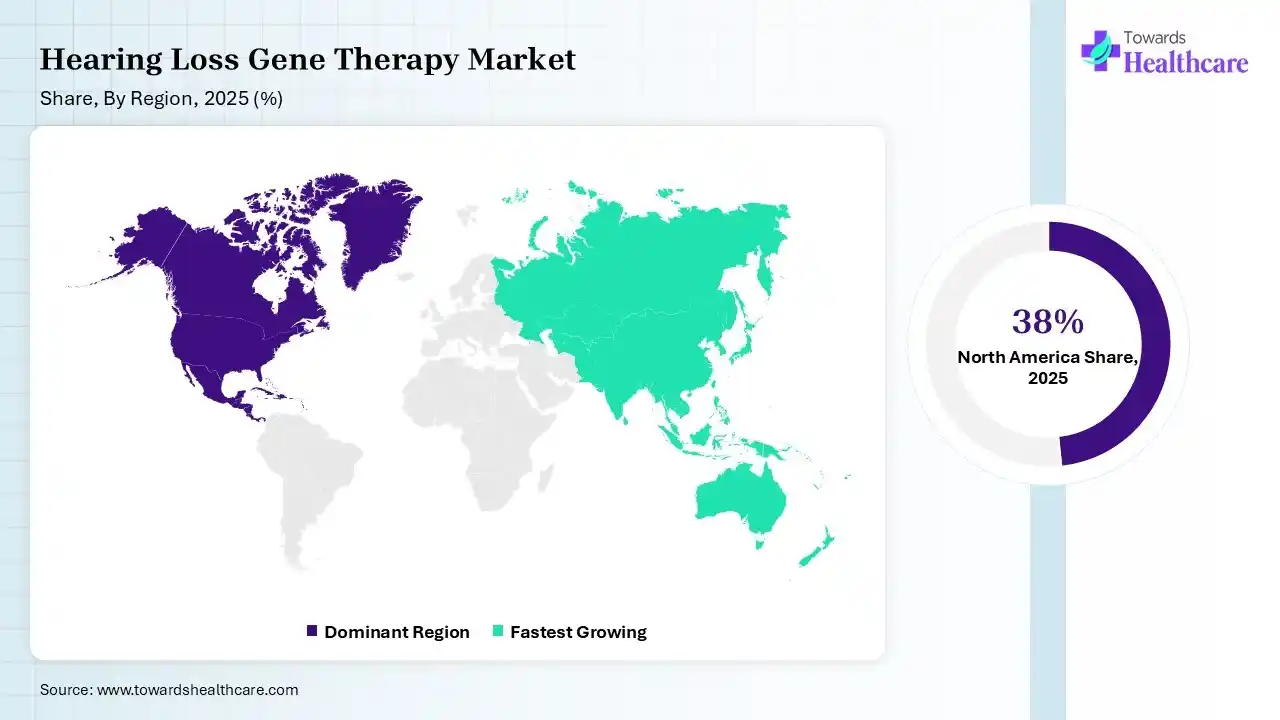
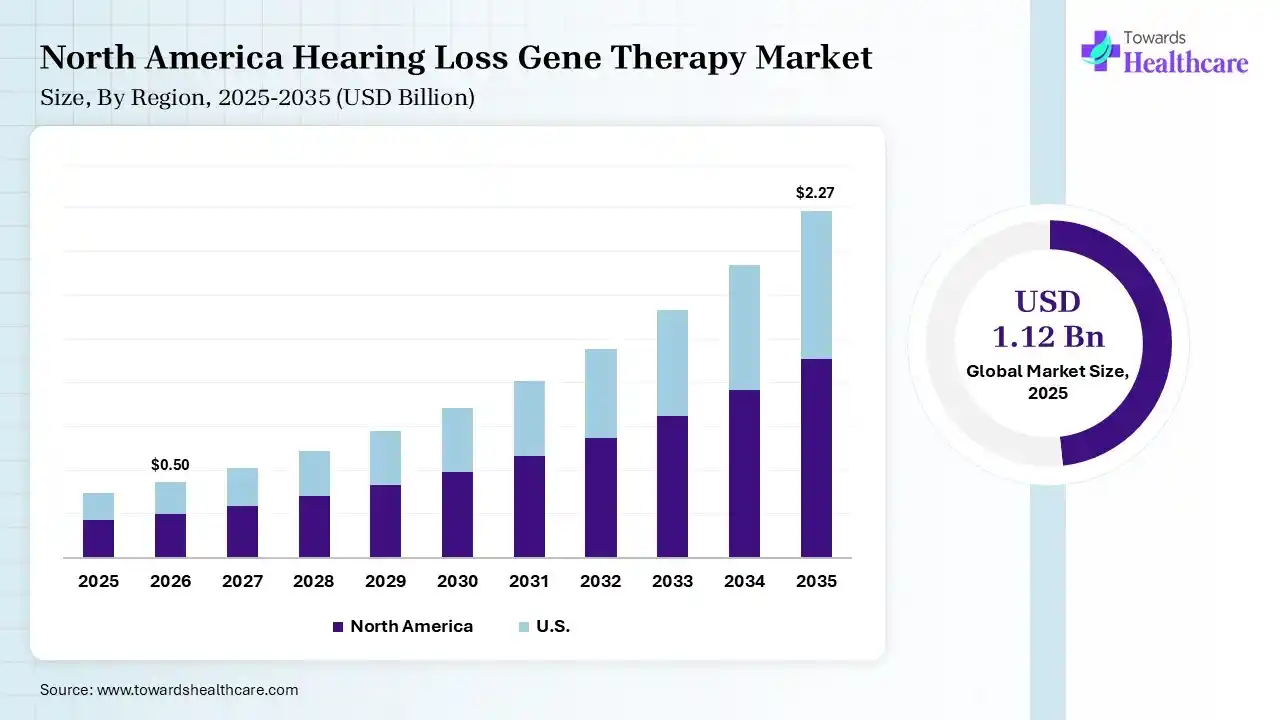
In 2025, North America led with a 38% share of the hearing loss gene therapy market, due to the strong presence of biotech leaders, which surges innovation. Also, the region has a well-nurtured healthcare infrastructure, which supports the broader adoption of these therapies. Additionally, the regional major private & public funding is spurring clinical trials for therapies, with a focus on restoration of hair cell function.
U.S. Market Trends
In the U.S., ongoing clinical trials are often targeting DFNB9, a specific form of congenital auditory neuropathy, coupled with prospective research activities on other genes, like TMPRSS3, PCDH15, & TMC1. Moreover, the respective market is demonstrating early studies, which show successful improvements in hearing & speech in children.
Asia Pacific held 23% of the market share and is predicted to expand rapidly in the hearing loss gene therapy market. Major drivers involved in the upcoming progress are the vast geriatric population, mainly in China & Japan, which are highly prone to sensorineural hearing loss. This population is increasingly demanding the latest solutions for this type of concern. Many governments are heavily investing in biotechnology & gene therapy research by offering grants & easy approval measures.
China Market Trends
Moreover, China is estimated to grow at a rapid CAGR in the coming era. Specifically, teams from Fudan University's Eye & ENT Hospital are expanding globally in OTOF-related deafness (DFNB9) gene therapy. Numerous large-scale Chinese trials are developing rapidly to assess infants, children, & adults, with 90% of participants having substantial hearing optimization.
R&D
Clinical Trials & Regulatory Approvals
Patient Support & Services
| Company | Description |
| Regeneron Pharmaceuticals | It has unveiled Otarmeni (lunsotogene parvec-cwha), the first-ever FDA-approved gene therapy for genetic hearing loss. |
| Akouos (Eli Lilly) | This firm leads in the development of Adeno-associated viral (AAV) vector-based gene therapies for inner ear issues. |
| Sensorion | Its groundbreaking portfolio emphasizes SENS-501 for OTOF gene-mediated deafness. |
| Fudan University | This mainly provides gene therapy treatments & clinical trials for hereditary hearing loss, especially for the rare condition DFNB9. |
| Otovia Therapeutics | It has explored OTOV-101, an innovative dual adeno-associated virus (AAV) gene therapy. |
| Astellas Pharma | This company facilitates a variety of regenerative medicine & in vivo gene therapy solutions to repair inner ear hair cells to treat sensorineural hearing loss. |
| BridgeBio Pharma | Its offerings cover BBP-815, an investigational adeno-associated virus (AAV) gene therapy. |
Strengths
Weaknesses
Opportunities
Threats
By Therapy Type
By Vector Type
By Hearing Loss Type
By Delivery Method
By End User
By Region
May 2026
May 2026
April 2026
April 2026