April 2026

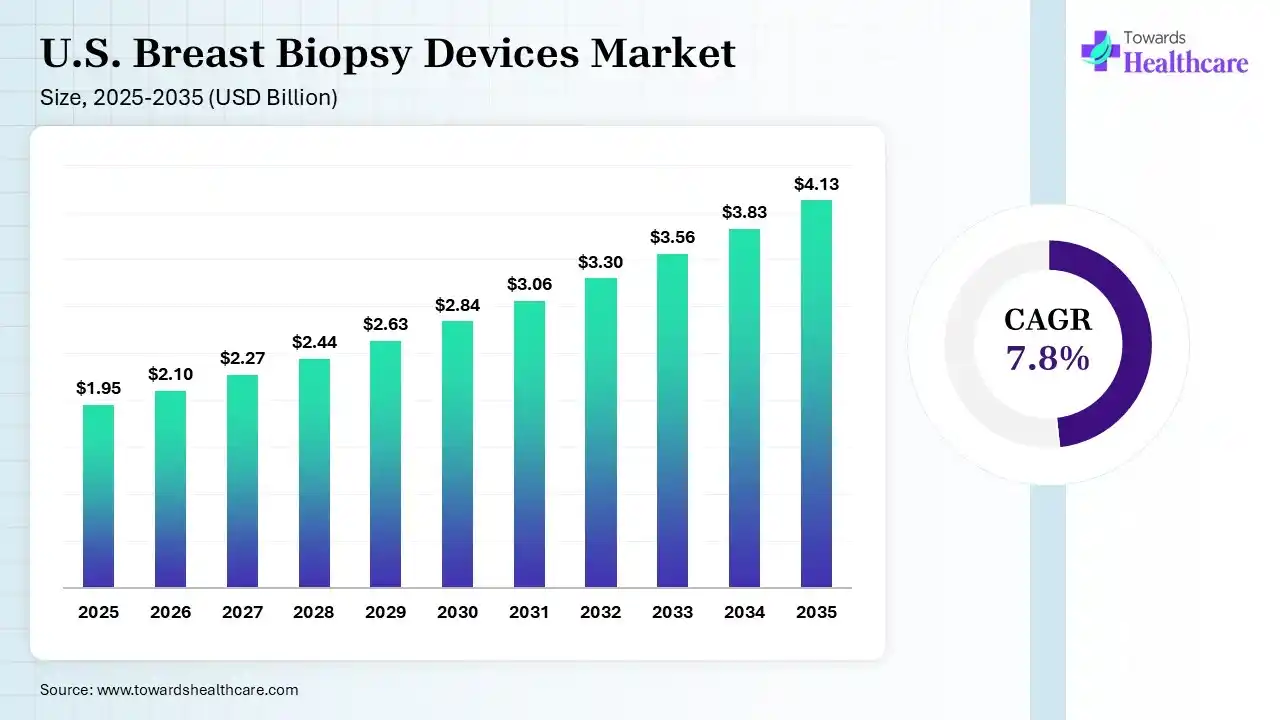
The U.S. breast biopsy devices market size was estimated at USD 1.95 billion in 2025 and is predicted to increase from USD 2.1 billion in 2026 to approximately USD 4.13 billion by 2035, expanding at a CAGR of 7.8% from 2026 to 2035. The market is growing as more women undergo screening and hospitals adopt advanced, minimally invasive diagnostic technologies. Demand is rising for accurate, image-guided procedures, driving steady market expansion in the coming years.
Breast biopsy devices are medical tools used to detect tissue samples from the breast for detecting cancer or other abnormalities, often using minimally invasive and image-guided techniques. The U.S. breast biopsy devices market is growing due to increasing breast cancer cases and heightened awareness about early detection. Hospitals and diagnostic centers are adopting a minimally invasive, image-guided biopsy technique for faster and more accurate results. Technological advancements, along with rising routine screening and preventive healthcare initiatives, are further driving demand, boosting market expansion steadily across the country.
Artificial Intelligence can revolutionize the market by enhancing imaging analysis, improving lesion detection, and guiding precise needle placements. AI-driven tools can reduce diagnostic errors, shorten procedure times, and personalize biopsy strategies based on patient data. Integration of machine learning with minimally invasive systems promises higher accuracy, better workflow efficiency, and faster clinical decision-making, accelerating adoption across hospitals and diagnostic centers.
| Table | Scope |
| Market Size in 2026 | USD 2.1 Billion |
| Projected Market Size in 2035 | USD 4.13 Billion |
| CAGR (2026 - 2035) | 7.8% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product, By Technology, By Guidance Technique, By End User, By Application |
| Top Key Players | Hologic, Inc., Mammotome (Danaher), BD (Becton Dickinson), Argon Medical Devices, Merit Medical Systems, GE HealthCare |
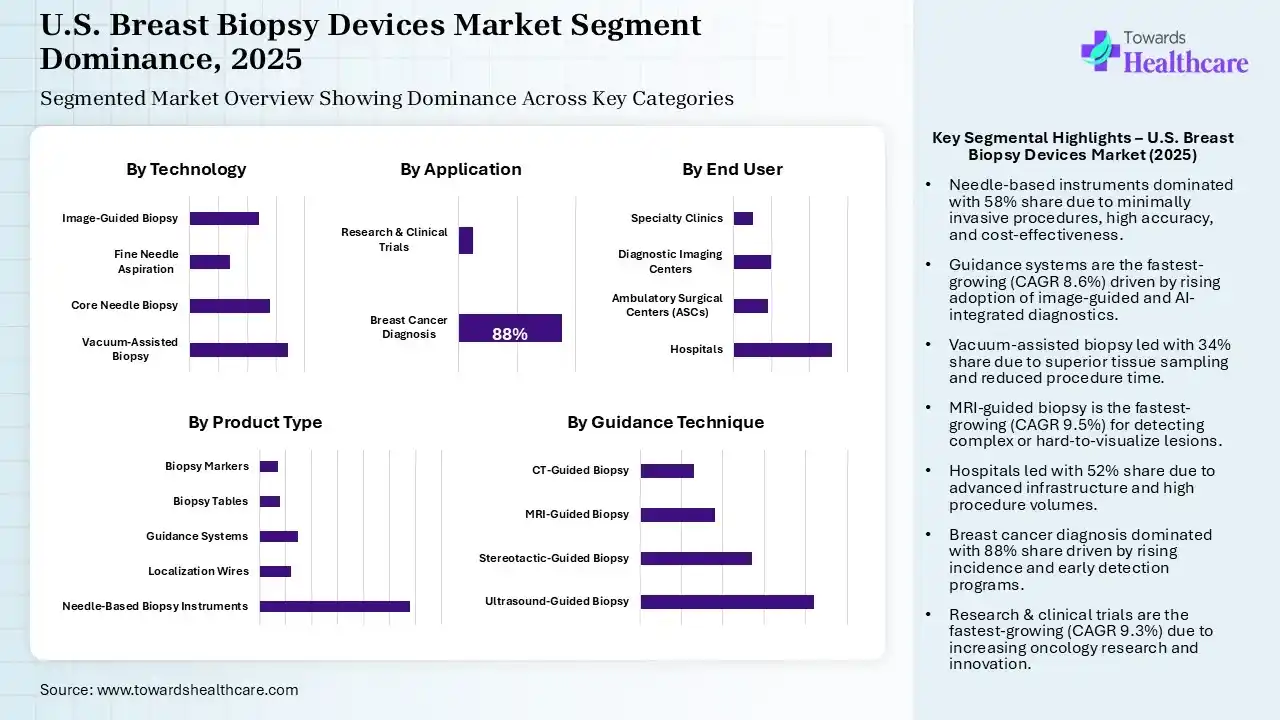
The Needle-Based Instruments Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Needle-Based Biopsy Instruments | 58% |
| Localization Wires | 12% |
| Guidance Systems | 15% |
| Biopsy Tables | 8% |
| Biopsy Markers | 7% |
The needle-based instruments segment dominated the U.S. breast biopsy devices market with shares of 58% in 2025 due to their minimally invasive nature, ease of use, and high diagnostic accuracy. Core-needle systems allow precise tissue sampling with reduced patient discomfort, shorter procedure times, and faster recovery. Their widespread availability in hospitals and diagnostic centers, along with cost-effectiveness compared to surgical biopsies, reinforced their market dominance.
The guidance systems segment held the second largest market share of 15% in 2025 and is expected to grow at the fastest CAGR of 8.6% in the market during the forecast period due to increasing adoption of image-guided procedures. Technologies like ultrasound, MRI, and stereotactic guidance enhance biopsy accuracy, reduce complications, and support minimally invasive techniques. Rising demand for precise diagnostics, coupled with technological advancements and integration with AI, is driving strong market expansion during the forecast period.
The localization wires segment held 12% of the U.S. breast biopsy devices market share in 2025, as they help accurately mark non-palpable breast lesions for surgical removal. Surgeons and radiologists rely on these wires to improve procedural precision, reduce tissue damage, and ensure complete excision. Increasing breast cancer screening, rising adoption of preoperative localization techniques, and their cost-effectiveness compared to advanced imaging alternatives are driving steady growth in this segment across hospitals and outpatient centers.
The biopsy tables segment held 8% of the market share in 2025 as they provide stable, ergonomic platforms for performing breast biopsies with enhanced accuracy and patient comfort. Their integration with imaging systems like mammography and ultrasound allows precise needle guidance. Rising procedure volumes, growing adoption of minimally invasive techniques, and demand for streamlined workflows in hospitals and diagnostic centers are driving steady growth in this segment across the U.S. market.
The Vacuum-Assisted Biopsy Segment Led the Market in 2025 with the Largest Share
| Segment | Share 2025 (%) |
| Vacuum-Assisted Biopsy | 34% |
| Core Needle Biopsy | 28% |
| Fine Needle Aspiration | 14% |
| Image-Guided Biopsy | 24% |
The vacuum-assisted biopsy segment led the U.S. breast biopsy devices market with shares of 34% in 2025 due to its ability to collect larger tissue samples with fewer needle insertions, improving diagnostic accuracy. Its minimally invasive approach reduces patient discomfort, shortens procedure time, and lowers complication risks. Increasing preference for precise, image-guided procedures in hospitals and outpatient centers, along with technological advancements in vacuum-assisted systems, has driven widespread adoption and market leadership in 2025.
The core needle biopsy segment held the second largest market share of 28% in 2025 due to its high diagnostic accuracy and minimally invasive nature. It allows precise tissue sampling with reduced patient discomfort and faster recovery compared to surgical biopsies. Widely available in hospitals and outpatient centers, its cost-effectiveness, reliability, and compatibility with image-guided procedures have fueled consistent adoption, making it a key contributor to the market.
The image-guided biopsy segment held 24% of the U.S. breast biopsy devices market share in 2025 and is expected to grow at the fastest CAGR of 9.20% in the market during the forecast period due to increasing demand for precise, minimally invasive diagnostics. Techniques using ultrasonography, MRI, and stereotactic guidance improve lesion localization, reduce procedural errors, and enhance patient safety. Rising breast cancer screening, technological advancements, and integration with AI-driven tools are driving adoption across hospitals and outpatient centers, fueling rapid market expansion during the forecast period.
The fine needle aspiration segment held 14% of the market share in 2025 due to its simplicity, low cost, and minimally invasive approach for extracting cells from breast lesions. It enables quick diagnosis with minimal discomfort and faster recovery. Increasing routine breast screening preference for outpatient procedures, and the need for rapid preliminary results, are driving its adoption, making FNA an increasingly important segment in the market.
The Ultrasound-Guided Biopsy Segment held a dominant position in the Market in 2025
| Segment | Share 2025 (%) |
| Ultrasound-Guided Biopsy | 42% |
| Stereotactic-Guided Biopsy | 27% |
| MRI-Guided Biopsy | 18% |
| CT-Guided Biopsy | 13% |
The ultrasound-guided biopsy segment held a dominant position in the U.S. breast biopsy devices market with a revenue share of 42% in 2025 due to the real-time imaging, high accuracy, and minimally invasive nature. It allows precise needle placements, reduces procedure time, and enhances patient comfort. Widespread availability in hospitals and diagnostic centers, cost-effectiveness, and growing preference for image-guided procedures for early breast cancer detection have reinforced its leadership in the market.
The stereotactic-guided biopsy segment held the second largest market share of 27% in 2025 due to its precision in targeting non-palpable breast lesions using mammography-based imaging. It allows minimally invasive tissue sampling with accuracy, reduces complications, and is widely adopted in hospitals and outpatient centers. Growing breast cancer screening, demand for accurate diagnostics, and compatibility with core and vacuum-assisted systems are driving steady growth in the segment.
The MRI-guided biopsy segment held 18% of the U.S. breast biopsy devices market share in 2025 and is expected to grow at the fastest CAGR of 9.50% in the market during the forecast period due to its ability to detect and target lesions that are difficult to visualize with other imaging techniques. Its high precision and minimally invasive approach improve diagnostic accuracy and patient outcomes. Increasing use of advanced imaging in high-risk patients and rising demand for accurate, image-guided procedures are driving rapid adoption in hospitals and diagnostic centers.
The CT-guided biopsy segment held 18% of the U.S. breast biopsy devices market share in 2025 due to its ability to accurately target deep or complex breast lesions that are difficult to detect with other imaging methods. Its precision, minimally invasive approach, and growing adoption in hospitals and diagnostic centers for high-risk patients are driving steady market expansion in the U.S. breast biopsy devices sector.
The Hospitals Segment Dominated the Market in 2025 with the Largest Share
| Segment | Share 2025 (%) |
| Hospitals | 52% |
| Ambulatory Surgical Centers (ASCs) | 18% |
| Diagnostic Imaging Centers | 20% |
| Specialty Clinics | 10% |
The hospitals segment dominated the U.S. breast biopsy devices market with a share of 52% in 2025 due to their advanced infrastructure, availability of specialized imaging equipment, and skilled medical professionals. Hospitals perform a high volume of diagnostic procedures, including minimally invasive and image-guided biopsies, ensuring accurate and timely results. Rising breast cancer screenings, comprehensive patient care, and preference for hospital-based procedures have reinforced their leading role in driving market growth in 2025.
The diagnostic imaging centers segment held the second-largest share of 20% of the market in 2025 and is expected to grow at the fastest CAGR of 9.00% in the market during the forecast period due to increasing preference for outpatient procedures, faster scheduling, and cost-effective services. These centers are adopting advanced, minimally invasive, and image-guided biopsy technologies, enhancing procedural efficiency and patient comfort. Rising breast cancer screenings and growing awareness of early detection are driving demand, positioning imaging centers as a rapidly expanding end-user segment in the market.
The ambulatory surgical centers (ASCs) segment held 18% of the U.S. breast biopsy devices market share in 2025 due to a shift in breast biopsy procedures to outpatient settings for convenience and cost efficiency. ASCs offer shorter wait times, streamlined workflows, and minimally invasive procedures with faster recovery. Rising patient preference for outpatient of advanced biopsy technologies in ASCs is fueling steady growth, making this segment an important contributor to the market.
The specialty clinics segment held 10% of the market share in 2025 because these centers focus on breast health and early detection, providing minimally invasive and image-guided biopsy procedures. Growing patient preference for specialized, convenient care, combined with rising awareness and routine screening, is driving adoption, making speciality clinics an increasingly important contributor to the market.
The Breast Cancer Diagnosis Segment held the Largest Market Share in 2025
| Segment | Share 2025 (%) |
| Breast Cancer Diagnosis | 88% |
| Research & Clinical Trials | 12% |
The breast cancer diagnosis segment held the largest market share of 88% in 2025, as early and accurate diagnosis is critical for effective treatment. Increasing breast cancer incidence, widespread screening programs, and rising awareness about early detection are driving demand for minimally invasive and image-guided biopsy demand for minimally invasive and image-guided biopsy procedures. hospitals and diagnostic centers prioritize these technologies to ensure precise tissue sampling, improving patient outcomes, and reinforcing the segment’s leading position in 2025.
The research & clinical trials segment held 12% of the U.S. breast biopsy devices market share in 2025 and is expected to grow at the fastest CAGR of 9.30% in the market during the forecast period due to increasing use of breast biopsy devices in developing new diagnostic techniques and therapies. Rising investment in oncology research, expanding clinical studies, and the need for precise tissue sampling are driving adoption, fueling rapid market growth during the forecast period.
According to the U.S. National Cancer Institute (SEER), an estimated 316,950 new female breast cancer cases are expected in 2025, highlighting strong demand for breast biopsy procedures and related diagnostic devices across the U.S. market.
R&D
Clinical Trials
Packaging and Serialization

| Companies | Headquarters | Offerings |
| Hologic, Inc. | Massachusetts, USA | provides minimally invasive and image-guided breast biopsy systems, specimen imaging tools, and integrated breast health solutions |
| Mammotome (Danaher) | Ohio, USA | specializes in vacuum-assisted biopsy devices and lesion localization solutions for hospitals and imaging centers. |
| BD (Becton Dickinson) | New Jersey, USA | offers vacuum-assisted and multi-modality biopsy systems, consumables, and precision interventional tools. |
| Argon Medical Devices | Texas, USA | delivers core needle and semi-automatic biopsy instruments along with localization products for breast procedures. |
| Merit Medical Systems | Utah, USA | provides core needle and automated biopsy devices designed for accurate image-guided sampling. |
| GE HealthCare | Illinois, USA | offers advanced imaging systems like mammography, MRI, and ultrasound that support breast biopsy procedures and diagnostics. |
Strengths
Weaknesses
Opportunities
Threats
By Product
By Technology
By Guidance Technique
By End User
By Application
April 2026
April 2026
April 2026
March 2026