April 2026

The global drug delivery nanoparticles formulation market size was estimated at USD 92.6 billion in 2025 and is predicted to increase from USD 105.38 billion in 2026 to approximately USD 337.31 billion by 2035, expanding at a CAGR of 13.8% from 2026 to 2035. The drug delivery nanoparticles formulation market is growing because nanoparticulate drug delivery systems are becoming more widely regarded as an effective means of directing biological drugs.
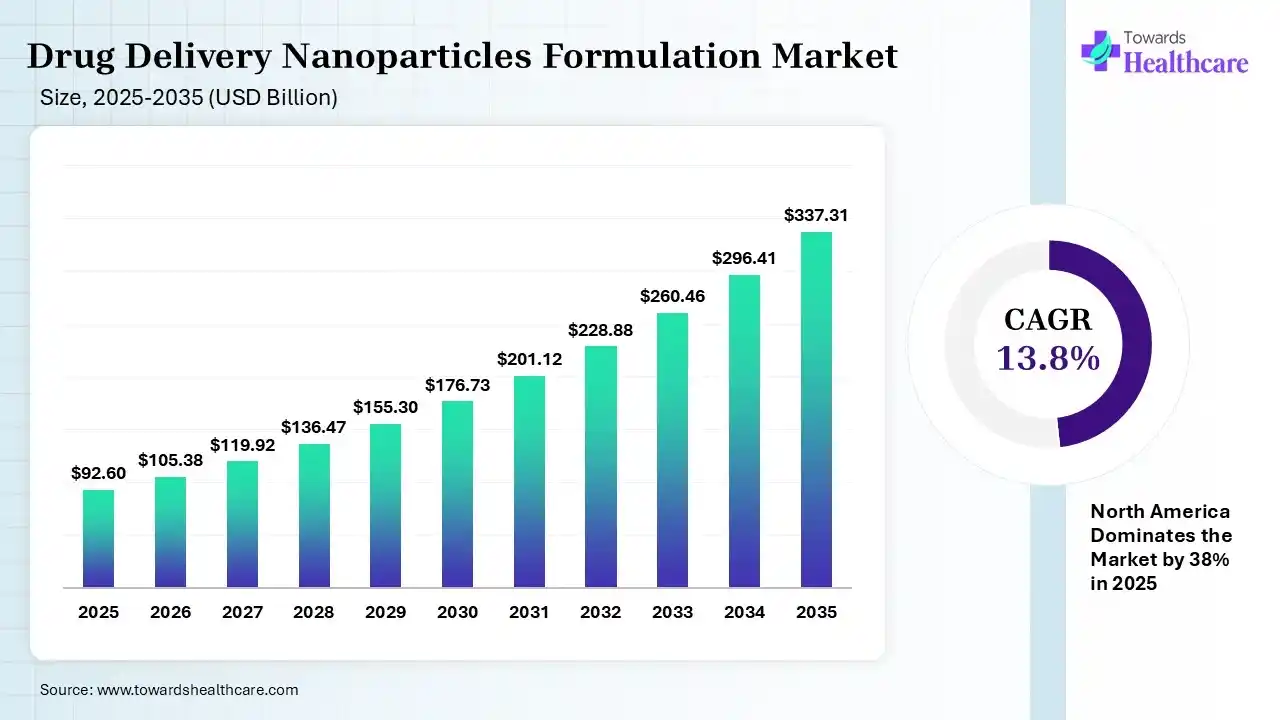
The drug delivery nanoparticles formulation market is growing as nanoparticle formulations are used for drug delivery in the brain and explored for imaging, sensing, and analytical determinations. Nanoparticle-driven drug delivery systems selectively deliver chemotherapeutic agents to cancer cells, reducing damage to healthy tissues and lowering side effects. Nanoparticles deliver drugs that promote angiogenesis, lower inflammation, or scavenge reactive oxygen species, therefore promoting tissue repair and regeneration. Nanoparticle-driven vaccines are evolving as a promising opportunity for improving immunization strategies. Nanomedicine’s potential advantages are significant; it is important to address the ethical and safety considerations related to using nanoparticles in drug delivery. Nanoparticles engineered to drive site-specific delivery, enhancing drug concentration at the intended site and reducing off-target effects.
Artificial intelligence (AI) is transforming nanoparticle (NP)-based drug delivery by enhancing design, synthesis, and optimization processes. AI and computational techniques can evaluate drug loading, retention, and formulation stability to enhance the compatibility between nanocarriers and drugs. Recent developments show that AI-based technology models exactly predict nanoparticle characteristics, optimize synthesis parameters, analyse challenging characterization information, and estimate biological performance, therefore reducing experimental workloads and speeding up development. These tools support better assessment of drug loading, retention, and formulation stability, ultimately refining nanocarrier drug pairing.
Smart & Stimuli-Responsive Carriers:
Stimuli-responsive nanoparticles (NPs) are a significant enhancement to standard drug delivery processes as they allow the release of therapeutic agents in response to a s biological response.
Predictive Analytics:
Predictive analytics uses statistical modelling, machine learning, and AI-based technology to forecast future trends, behaviours, or results. It supports researchers to optimize research designs, anticipate patient results, and allows businesses to make data-driven decisions, lower risks, and identify opportunities.
Personalized Nanomedicine:
Nanomedicine drug formulation could efficiently render formulated drugs stealthy to metabolizing enzymes, also make them intracellular in the endocytic process, which is independent of the transporter.
| Table | Scope |
| Market Size in 2026 | USD 105.38 Billion |
| Projected Market Size in 2035 | USD 337.31 Billion |
| CAGR (2026 - 2035) | 13.8% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Nanoparticle Type, By Drug Type, By Application, By Route of Administration, By End User, By Region |
| Top Key Players | Well Shield & Crystalplex, TRANSFERRA Nanosciences Inc., DIANT Pharma, Ipsen & Insmed, Bristol-Myers Squibb |
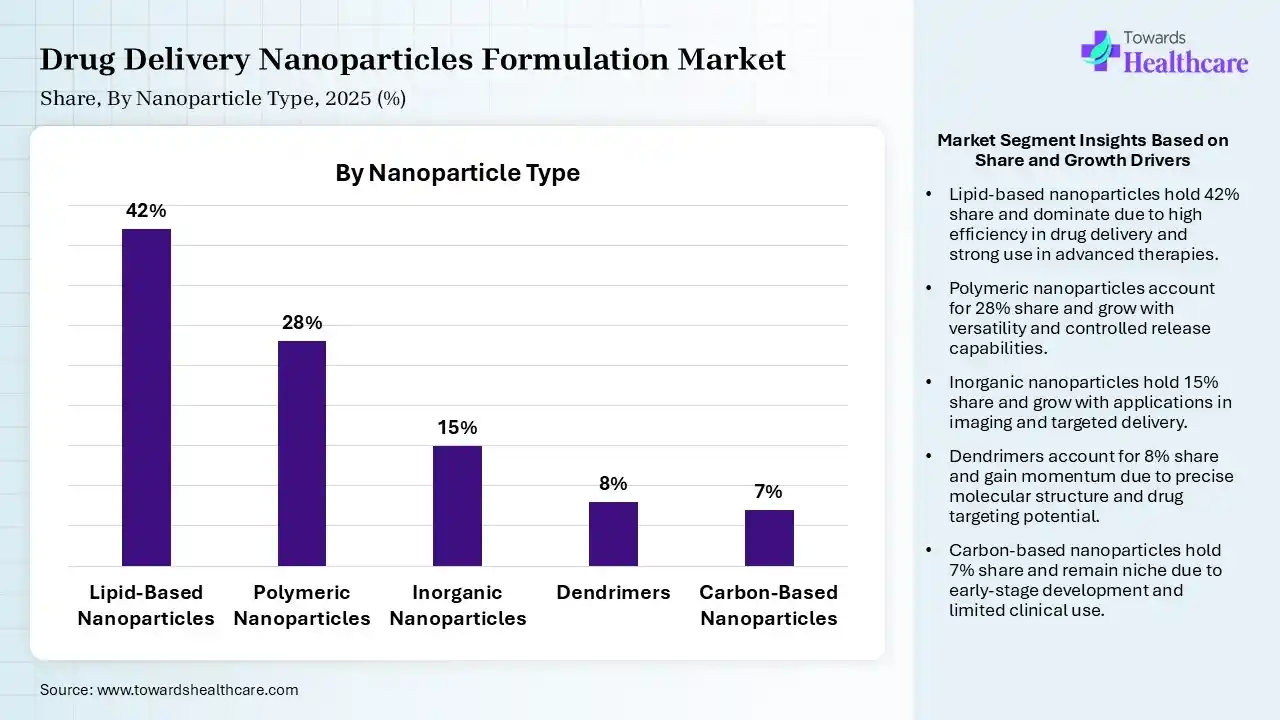
| Segment | Share 2025 (%) |
| Lipid-Based Nanoparticles | 42% |
| Polymeric Nanoparticles | 28% |
| Inorganic Nanoparticles | 15% |
| Dendrimers | 8% |
| Carbon-Based Nanoparticles | 7% |
The Lipid-Based Nanoparticles Segment Led the Drug Delivery Nanoparticles Formulation Market in 2025
The lipid-based nanoparticles segment contributed the largest market share of 42% in 2025, as they offer advantages such as high temporal and thermal stability, high loading capacity, ease of preparation, low production costs, and suitability for large-scale industrial production, since they are prepared from natural sources. LBNPs have been widely assayed in in vitro cancer therapy but also in vivo, with hopeful results in some clinical trials.
The polymeric nanoparticles segment held a significant share of 28% in the market, as polymeric NPs as drug carriers include their potential use for controlled release, the ability to protect drugs and other molecules with biological activity against the environment, and improve their bioavailability and therapeutic index. Polymeric nanoparticles (PNPs) can encapsulate a variety of therapeutic agents, enhance bioavailability, and facilitate controlled release, thereby improving therapeutic efficacy and minimizing side effects.
The inorganic nanoparticles segment held a significant share of 15% in the market, as these nanoparticles do have intrinsic outstanding physicochemical characteristics such as magnetic, thermal, optical, and catalytic performance, and therefore, these nanosized materials provide a sturdy framework where two or more dopants can be combined to give multifunctional capabilities. Inorganic nanoparticles are non-toxic, hydrophilic, biocompatible, and highly stable as compared to organic materials.
The dendrimers segment held a significant share of 8% in the market and is expected to grow at the fastest CAGR during the forecast period, as it provides major advantages of dendrimers over conventional antibiotics, such as their lower likelihood of inducing resistance. Dendrimers are valuable as transdermal and topical drug delivery systems for nonsteroidal anti-inflammatory drugs (NSAIDs), antiviral, anticancer, antimicrobial, and antihypertensive drugs.
The carbon-based nanoparticles segment held a significant share of 7% in the market, as carbon nanoparticles might play a noteworthy role in future agricultural results, involving the enhancement in food production and assistance with agricultural sustainability. CNP-driven material has high electrical conductivity, and the CNP transistor enables high amplification of cardiac electrical signals.
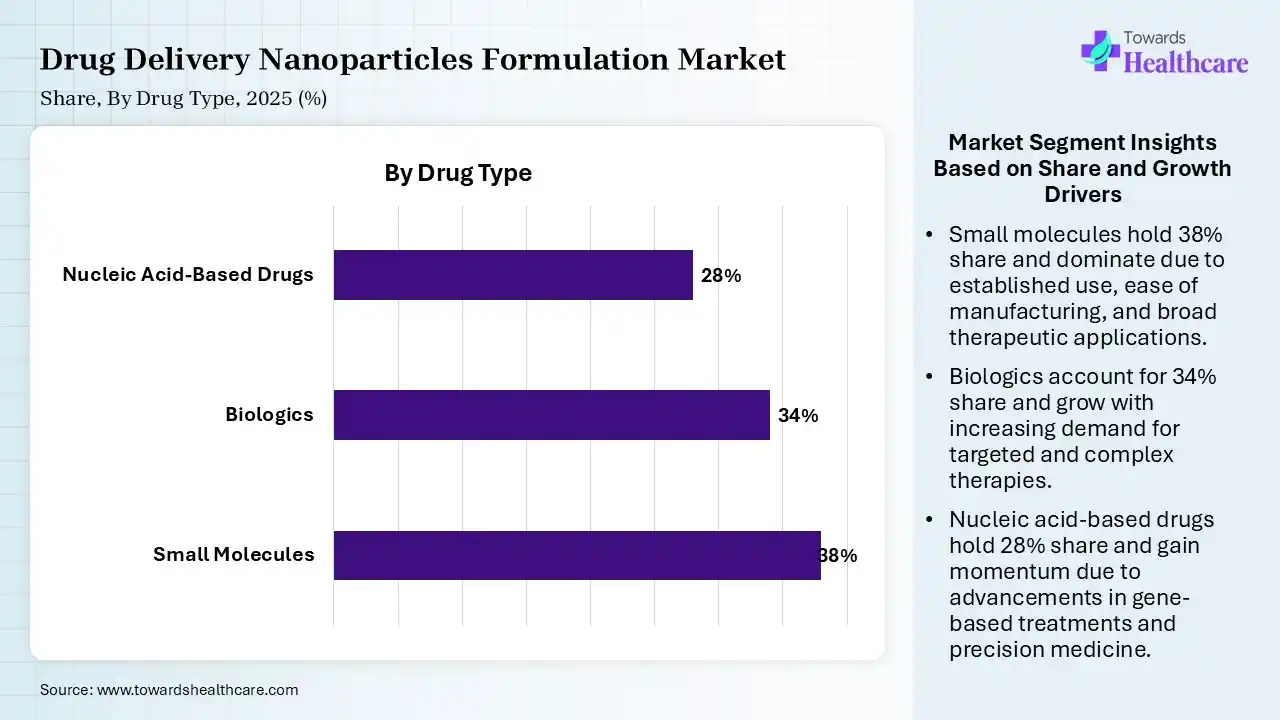
| Segment | Share 2025 (%) |
| Small Molecules | 38% |
| Biologics | 34% |
| Nucleic Acid-Based Drugs | 28% |
Small Molecules Segment Led the Drug Delivery Nanoparticles Formulation Market in 2025
The small molecules segment contributed the largest market share of 38%, as small molecules are processed into easy-to-ingest tablets or capsules. Small structure and chemical composition lead to the easier penetration of small molecules into cell membranes or target organs, presenting intracellular benefits compared to extracellularly directed, highly specific biological proteins.
The biologics segment held a significant share of 34% of the market in 2025. Biodegradable polymers such as PLGA allow sustained release over weeks or months, lowering the frequency of injections. Advanced systems minimize off-target effects and systemic toxicity by delivering the therapeutic directly to the disease site. Biologics formulated as a dry powder are more stable and are less susceptible to degradation.
The nucleic acid-based drugs segment held a significant share of 28% of the market, and is expected to grow at the fastest CAGR during the forecast period, as nucleic acid drugs control the biological functions of cells, based on nucleotide sequence information. Nucleic acid drugs (NADs) are a novel generation of gene-editing modalities considered by their increasing efficiency and rapid advancement.
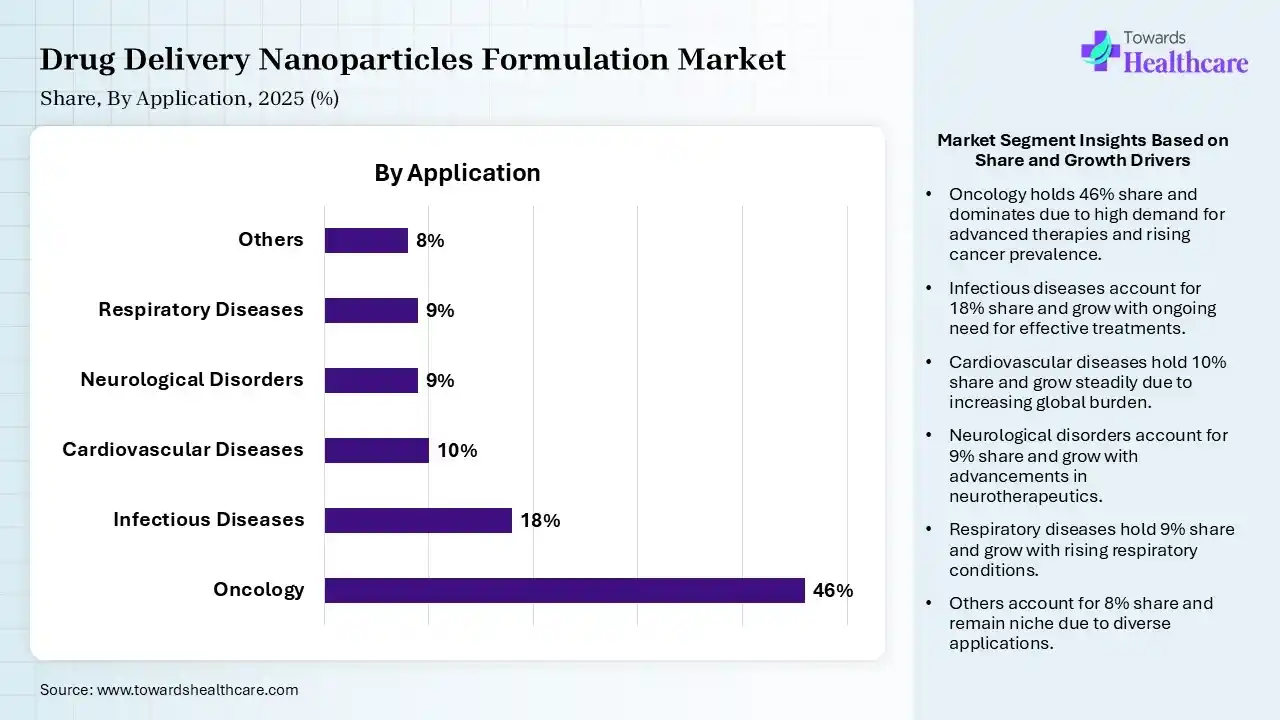
| Segment | Share 2025 (%) |
| Oncology | 46% |
| Infectious Diseases | 18% |
| Cardiovascular Diseases | 10% |
| Neurological Disorders | 9% |
| Respiratory Diseases | 9% |
| Others | 8% |
Oncology Segment Led the Drug Delivery Nanoparticles Formulation market in 2025
The oncology segment contributed the largest market share of 46% in 2025, as nano drug-delivery systems for natural anticancer agents enhance bioavailability, biodistribution, therapeutic activity, drug targeting, and stability. Nucleic acids have many advantages as nanomaterials, like high biocompatibility, low immunogenicity, design flexibility, small size, relatively low price, and simple functionalization.
The infectious diseases segment held a significant share of 18% in the market, as its capability to lower the rate of antigenic degradation, improve the stability of antigens, immunogenicity, and improve vaccine therapeutic effectiveness, drives efficient phagocytosis and rapid processing by improving cellular membrane penetrability and APCs
The cardiovascular diseases segment held a significant share of 10% in the market, as nanoparticle drug delivery systems are used to enhance the safety and effectiveness by passive or active targeted delivery and to enhance the solubility of drug candidates. Nanoparticles proved their role as an efficient and reliable platform for the controlled and targeted delivery of drugs to cure lipid disorders, inflammation, thrombosis, and angiogenesis in atherosclerotic plaques.
The neurological disorders segment held a significant share of 9% in the market, and is expected to grow at the fastest CAGR during the forecast period, as nanoparticle delivery systems emerged as a significant strategy for driving the passage of siRNA over the blood–brain barrier for AD therapy. Nanoparticle drug delivery systems have evolved as an advancing strategy in managing central nervous system disorders.
The respiratory diseases segment held a significant share of 9% in the market, as nano-based drug delivery systems improve solubility, extend drug half-life, and achieve localized accumulation in the lungs by penetrating mucosal barriers. Using nanoparticles to deliver drugs involves an enlarged drug concentration at the disease site, minimised drug degradation, and loss. Nanoparticles provide targeted drug delivery for the efficient treatment of pulmonary diseases.
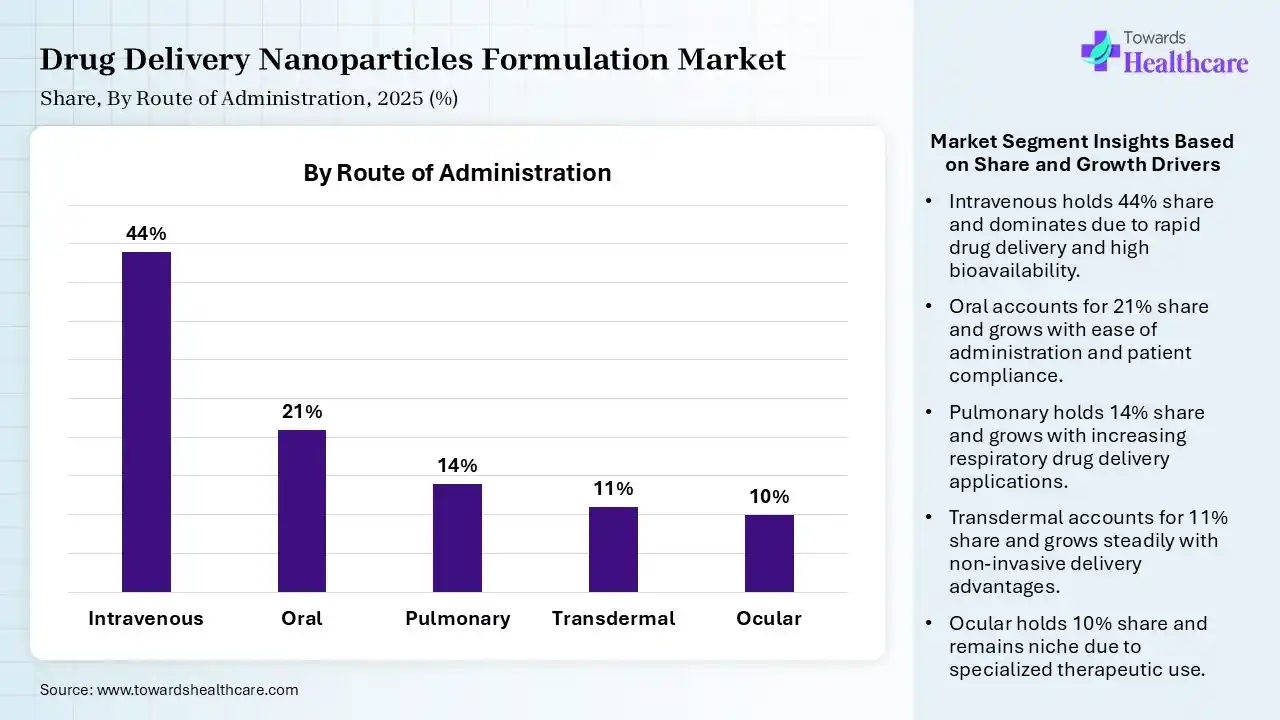
| Segment | Share 2025 (%) |
| Intravenous | 44% |
| Oral | 21% |
| Pulmonary | 14% |
| Transdermal | 11% |
| Ocular | 10% |
Intravenous Segment Led the Drug Delivery Nanoparticles Formulation Market in 2025
The intravenous segment contributed the largest market share of 44%, as intravenous injection is the most significant parental route of medication administration and bypasses the liver's first-pass metabolism. Intravenous medicines deliver a fast-acting therapeutic effect immediately, which is important in developing situations, like cardiac arrest or narcotic overdose. They are valuable for managing pain and nausea by rapidly achieving therapeutic levels.
The oral segment held a significant share of 21% in the market, and is expected to grow at the fastest CAGR during the forecast period, as oral administration of medication is an affordable, suitable, and most commonly used medicine administration route. It is significant site of drug absorption is generally the small intestine, and the bioavailability of the therapeutic is influenced by the quantity of drug absorbed in the intestinal epithelium.
The pulmonary segment held a significant share of 14% in the market, as pulmonary drug delivery has the advantage of being fast, efficient, and well-targeted, with rare systemic side effects. Pulmonary drug delivery provides a noninvasive, alternative technology to subcutaneous injection and intravenous injection. The pulmonary tract is inclined to be considered a promising and attractive route for the administration of active substances.
The transdermal segment held a significant share of 11% in the market, as this drug delivery process has major advantages over other routes of administration. Significant technology includes sustained release, bypassing first-pass metabolism, and improving patient compliance. It provides a controlled and sustained release of drugs, avoids first-pass metabolism in the liver, and lowers the potential for gastrointestinal adverse effects.
The ocular segment held a significant share of 10% in the market, as ocular drug administrations are preferred as it provides major advantages like avoiding the challenges of the enteral system, like first pass metabolism effect, growing retention time, and therapeutic effectiveness.
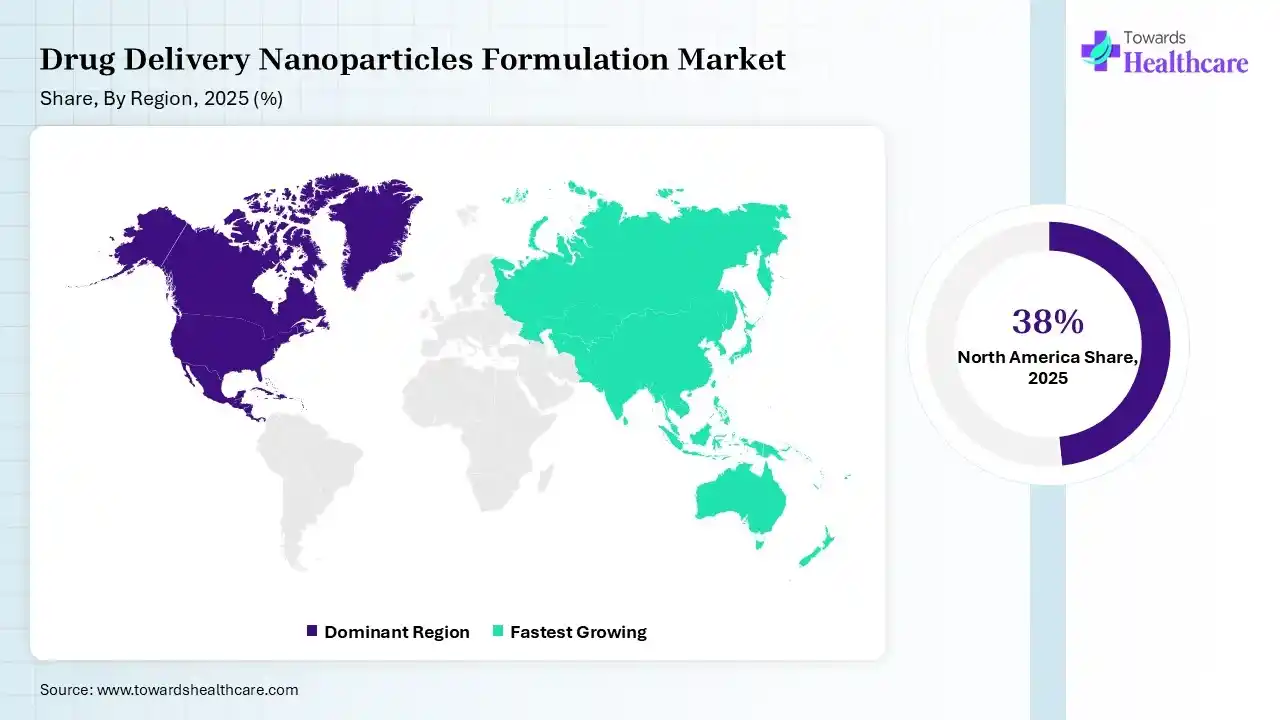
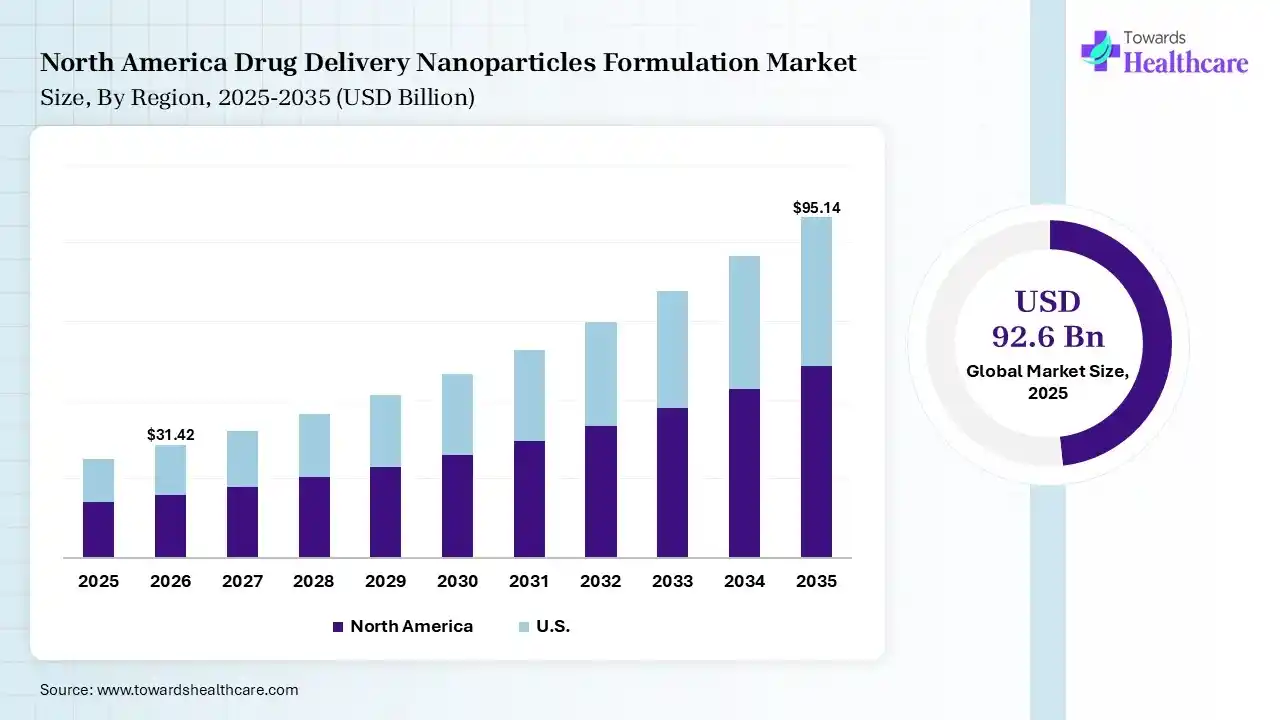
In 2025, North America dominated the drug delivery nanoparticles formulation market with a share of 38% in 2025, as pharmaceutical organizations have announced significant manufacturing spending. Presence of a biotech home with an increasing concentration of biotechnology organizations, research institutions, clinical trial infrastructure, and supporting organizations. Innovative pharmaceutical organizations make substantial spending in high-risk R&D. The academic institutions offer advanced research on disease mechanisms, while pharmaceutical organizations contribute advanced screening technology and drug development expertise, which contributes to the growth of the market.
U.S. Market Trends
The U.S. long-term productivity and economic growth. An increase in public infrastructure by itself increases the productivity of private capital, as public capital is a complement to private capital. Increasing government spending supports meet these capital requirements, reducing challenges and attracting private investors, thus offering support to industries significant to national security. U.S. organizations increasingly compete against firms backed by strong manufacturing policies
Asia Pacific held 24% share of the market, and is expected to have the fastest growth during the forecast period, as the increasing aging of the worldwide population exerts a substantial burden on neurological disorders, with aging predominantly prominent in Asia Pacific, which has the highest number of aging patients. The presence of large populations in China and India makes studying neurological disorders in the Asia Pacific imperative, which drives the growth of the market.
India Market Trends
India is presenting a similar trend since certain pharmaceutical organizations are shifting their attention towards NDDS advancement. Pharmaceutical organizations are focused on novel drug delivery systems to overcome the limitations of using conventional dosage forms. India’s increasing momentum in biopharma novelty highlights a significant shift from being mainly a generics powerhouse to becoming a home for advanced healthcare services.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Well Shield & Crystalplex | India | Researchers have developed ion-pair stealth shields that allow nanoparticles to bypass the body's immune defences. |
| TRANSFERRA Nanosciences Inc. | Canada | It remains to leverage Transferra's expertise in producing challenging lipid nanoparticles for RNA-driven therapies. |
| DIANT Pharma | United States | In September 2025, DIANT Pharma, Inc. and RNAV8 Bio announced a strategic partnership to provide biopharma organizations with an integrated path from mRNA sequence design via scalable lipid nanoparticle (LNP) manufacturing. |
| Ipsen & Insmed | United States | Ipsen has focused strongly on long-acting recombinant technology. |
| Bristol-Myers Squibb | New Jersey | Bristol Myers Squibb has agreed to acquire Orbital Therapeutics, a confidentially held biotechnology organization pioneering a novel generation of RNA medicines that reprogram the immune system in vivo. |
Strengths
Weaknesses
Opportunities
Threats
By Nanoparticle Type
By Drug Type
By Application
By Route of Administration
By End User
By Region
April 2026
April 2026
April 2026
February 2026