April 2026

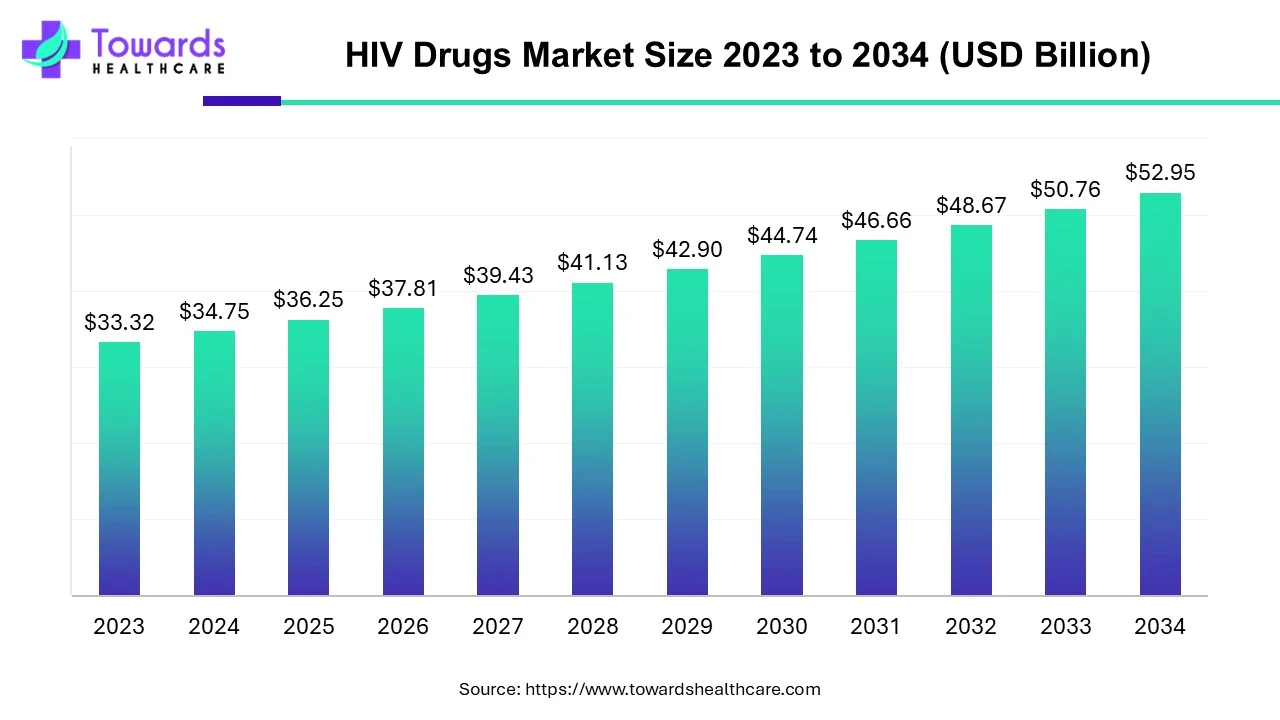
The HIV drugs market size is expected to grow from USD 36.25 billion in 2025 to USD 52.95 billion by 2034, with a CAGR of 4.3% throughout the forecast period from 2025 to 2034. The rising prevalence of HIV/AIDs, growing awareness for early diagnosis, and technological advancements drive the market.
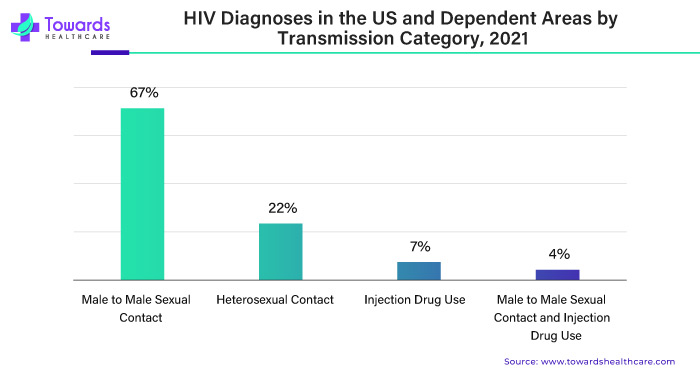
The World Health Organization estimated that around 39.9 million people were living with HIV at the end of 2023. This shows we need better treatments to fight HIV. HIV (Human Immunodeficiency Virus) weakens the body's immune system, making it vulnerable to infections and diseases. It is mainly transmitted through sharing needles or syringes, unprotected sexual contact, and from mother to child during childbirth or breastfeeding.
Early diagnosis of HIV is crucial because it allows for timely medical intervention and treatment. When HIV is diagnosed early, individuals can start antiretroviral therapy (ART) promptly. ART helps to suppress the virus, allowing the immune system to recover and preventing the progression of HIV to AIDS (Acquired Immunodeficiency Syndrome). Without treatment, HIV can lead to AIDS, a condition characterized by severe immune system damage and an increased risk of opportunistic infections and certain cancers. However, with proper medical care and treatment, people living with HIV can lead long and healthy lives.
Early detection plays a crucial role in preventing the transmission of HIV to others. Being aware of their HIV status enabled individuals to take necessary precautions, such as practicing safer sex and using sterile needles for injections. Additionally, early diagnosis facilitated access to support services and resources that aided individuals in dealing with the physical, emotional, and social challenges associated with HIV. These services encompassed counseling, support groups, and assistance with medication adherence.
HIV is a virus that attacks the immune system, leading to increased susceptibility to infections and diseases. Early diagnosis and treatment are essential for preventing the progression of HIV, improving health outcomes, and reducing the risk of transmission to others. Individuals must get tested regularly for HIV, especially if they engage in behaviors that may put them at risk of infection. With timely intervention and proper medical care, HIV can be managed effectively, allowing individuals to live fulfilling lives while reducing the impact of the virus on both individuals and communities.
Artificial intelligence (AI) streamlines the entire research process to develop novel drugs and drug delivery systems. It plays a vital role in screening and early diagnosis of HIV. It can assess a patient’s conditions and help researchers in developing personalized HIV drugs. AI can also identify the potential biomarkers and targets involved in disease progression. It can evaluate the interaction of novel drugs with the targets in silico. AI can also predict the physicochemical and pharmacokinetic properties of HIV medications, enabling healthcare professionals to assess the effects of these drugs on the body. Thus, the integration of AI into global health efforts offers a promising pathway to more efficient, effective, and equitable healthcare solutions.
The global number of people living with HIV/AIDS dramatically influences the demand for HIV medications, such as antiretroviral therapy (ART). As the number of people with HIV increases, so does the need for these drugs to manage the virus and maintain individuals' health.
As the number of people with HIV increases, more people will need access to treatment to help them live longer and healthier lives. These drugs don't just help individuals—they also help to slow down the spread of HIV in communities. When more people have access to HIV drugs, it can also help reduce the stigma surrounding HIV/AIDS. People are more likely to get tested and seek treatment if they know effective drugs are available. This can lead to earlier diagnosis and better outcomes for those living with HIV.
Furthermore, when more people are on treatment, it can lower the risk of transmitting the virus to others. This is because HIV drugs can reduce the amount of virus in a person's body, making them less likely to pass it on to someone else. As the number of people living with HIV/AIDS goes up, so does the need for HIV drugs. Ensuring that these drugs are available to everyone who needs them is crucial for controlling the spread of the virus and improving the health and well-being of individuals and communities worldwide.
Scientists are always working hard to find better ways to treat HIV. Their ongoing research helps them discover new and improved HIV drugs. These new drugs are often more effective at fighting the virus and keeping people healthy.
One way researchers make progress is by developing innovative drug delivery systems. These systems help get the drugs to where they're needed in the body more effectively. For example, some drugs might be made into long-acting injections that only need to be taken once a month instead of every day.
Combination therapies are another essential advancement. This means using multiple drugs together to attack the virus from different angles. By combining drugs, doctors can better control HIV and prevent it from becoming resistant to treatment. Long-acting formulations are also making a difference. Instead of having to take pills every day, some HIV drugs are being developed as injections or implants that only need to be administered every few weeks or months. This can make it easier for people to stick to their treatment plans.
These advancements in HIV treatment are helping to improve outcomes for people living with HIV. They make it easier for patients to take their medication as prescribed and can lead to better virus control. As research continues, we can expect even more progress in the fight against HIV/AIDS.
Combination therapy has been vital for making HIV drugs better and helping the HIV drug market grow. Instead of just using one drug, doctors now use a mix of drugs together to fight HIV. This combo approach is the usual way doctors treat HIV because it's more effective at stopping the virus and preventing it from becoming resistant to drugs.
| Sr. No. | Approved Date | Combination Therapy (Generic Name) | Combination Therapy (Brand Name) |
| 1. | March 30, 2022 | Abacavir/ Dolutegravir/ Lamivudine | Triumeq PD |
| 2. | January 22, 2021 | Cabotegravir and Rilpivirin | Cabenuva |
Each drug in the combo targets HIV in different ways, which makes it harder for the virus to spread. Because more people are getting this combo treatment, the demand for these drugs is going up. This means drug companies are making more HIV drugs to meet this need.
Combination therapy has also made treatment better for patients. Doctors can personalize treatment plans for each patient by using a mix of drugs. Plus, taking fewer pills each day as part of a combo treatment can make it easier for patients to stick to their treatment plans. Combination therapy has been a big reason why the HIV drugs market has grown. It's changed the way we treat HIV for the better, helping patients get better and fighting against HIV/AIDS.
HIV is a tricky virus because it can change and become resistant to the drugs used to treat it. This means that over time, the drugs may not work as well as they used to. So, scientists must keep working hard to create new medicines and treatment plans to fight against these changes. When HIV mutates, it can develop ways to escape the effects of the drugs, making them less effective. This is why constantly creating new treatments is essential to stay one step ahead of the virus.
Researchers study how HIV mutates and try to find new drugs that can still fight the virus even when it changes. They also look for ways to use different drugs together to make it harder for HIV to become resistant. Developing new drugs takes time and lots of testing to ensure they're safe and effective. But it's crucial for keeping HIV under control and helping people live longer and healthier lives.
Staying ahead of HIV's ability to mutate is a big challenge. Still, by continually developing new drugs and treatment strategies, scientists are working hard to overcome it and improve outcomes for people living with HIV.
By drug type, the nucleoside reverse transcriptase inhibitors (NRTIs) segment is predicted to witness significant growth in the market over the forecast period. Examples of NRTIs include abacavir, lamivudine, zidovudine, emtricitabine, and tenofovir. These drugs target the reverse transcriptase enzyme that converts viral RNA into DNA, preventing HIV from replicating. This prevents the further spread of the disease. It is the first line of treatment for HIV. The growing research and development activities lead to the development of novel NRTIs with enhanced efficacy and reduced side effects.
By distribution channel, the hospital pharmacies segment led the global market in 2024. The segmental growth is attributed to favorable infrastructure and suitable capital investments. Patients prefer hospitals for the treatment of HIV due to the presence of skilled professionals and multidisciplinary expertise. Hospital pharmacies contain updated and novel drugs based on the patient’s conditions.
By distribution channel, the retail pharmacies segment is projected to expand rapidly in the market in the coming years. Retail pharmacies offer specialized services, such as free home delivery, special discounts, and 24/7 facilities. The availability of generic alternatives attracts more patients to retail pharmacies. The increasing number of retail pharmacies potentiates the accessibility of medications. In the U.S., there is at least 1 pharmacy within 5 miles of households.
North America, particularly the United States, has a significant burden of HIV/AIDS. While the prevalence of HIV varies across different regions within North America, urban areas and specific populations, such as racial/ethnic minorities, bear a disproportionate burden of the disease.
Governments in North America, particularly in the United States and Canada, have implemented comprehensive HIV/AIDS prevention, treatment, and care programs. These initiatives include funding for research, prevention efforts, access to healthcare services, and subsidizing the cost of HIV drugs for eligible individuals. North America is home to several major pharmaceutical companies that develop, manufacture, and distribute HIV drugs. These companies play a pivotal role in the HIV drug market, driving innovation, conducting clinical trials, and bringing new medicines to market.
Governments in the Asia-Pacific region have implemented various initiatives to combat HIV/AIDS, including the expansion of treatment programs and the promotion of safe sex practices. These efforts contribute to the growth of the HIV drug market by increasing access to treatment and reducing the stigma surrounding the disease. Affordability is a significant factor influencing access to HIV drugs in the Asia-Pacific region. Many countries in this region have low to middle-income economies, and patients may struggle to afford expensive antiretroviral medications. Generic versions of HIV drugs and initiatives to reduce drug prices are essential for expanding access to treatment.
In January 2025, Blanver announced an investment of R$592 million in constructing its third HIV medication production plant. Sérgio Frangioni, CEO of Blanver, anticipated that these investments will double its revenue over three years from R$800 million to R$1.6 billion. He also said that to sustain growth, these investments were essential in all facilities. The new facility is expected to open by the end of 2025.
By Drug Type
By Distribution Channel
By Geography
April 2026
February 2026
February 2026
January 2026