May 2026

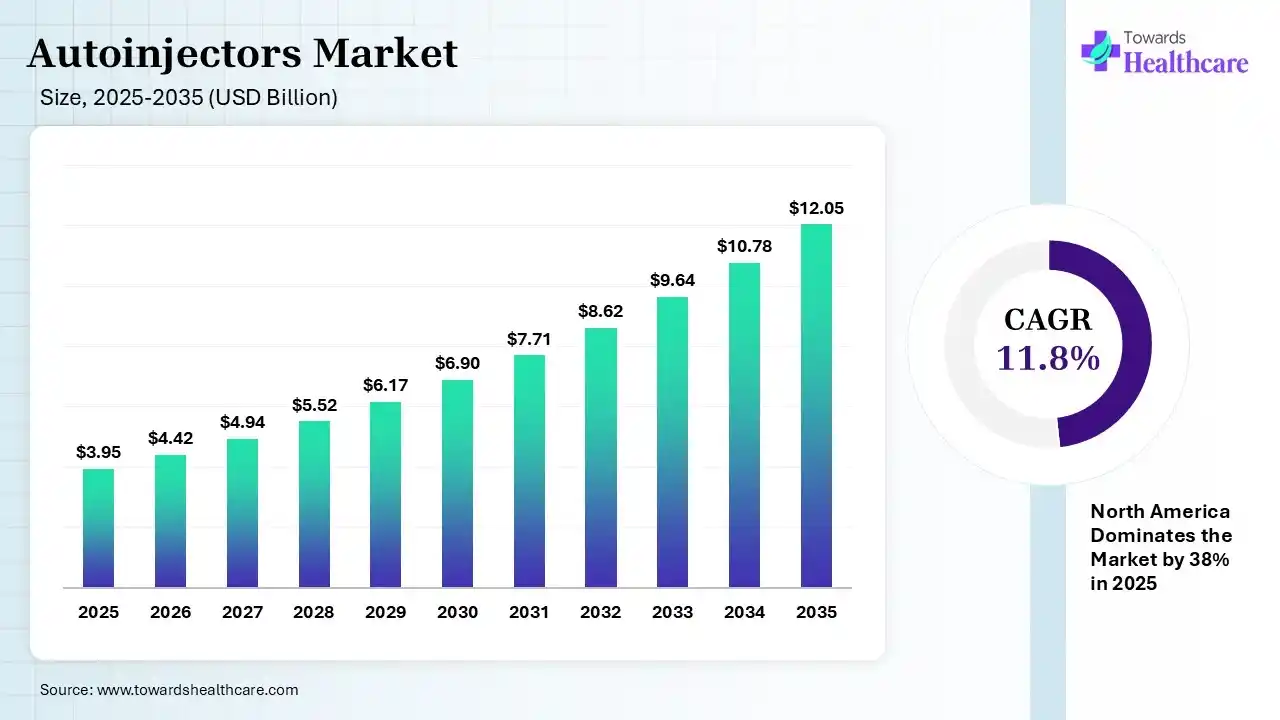
The global autoinjectors market size was estimated at USD 3.95 billion in 2025 and is predicted to increase from USD 4.42 billion in 2026 to approximately USD 12.05 billion by 2035, expanding at a CAGR of 11.8% from 2026 to 2035. The autoinjector market is growing because these tools increase patient compliance and adherence, as the drugs can be self-administered.
Autoinjectors are self-injectable tools; they are a significant class of healthcare devices that deliver drugs via subcutaneous or intramuscular injection. They enclose prefilled syringes and cartridges, which are driven by a spring system. This device is simple to self-administer, enhances patient compliance, lowers anxiety, and improves dosage accuracy. An autoinjector is an injection tool that allows the consumer to deliver a predetermined drug dosage via self-injection. An autoinjector is a single-dose product for pre-filled syringes with maximum fill volumes from 1 mL to 5.5 mL. Autoinjectors are often used in the military to protect personnel from chemical warfare agents. Autoinjector devices are significant for the rapid administration of drugs and antidotes, and they are used by those who have not been medically trained.
AI-based technology integration is transforming auto-injector stages, from enhancing patient compliance and experience to navigating challenging government landscapes. AI-based technology in drug discovery quickens target identification and molecule design, lowering R&D timelines. AI-based algorithms adapt to specific patient requirements, potentially adjusting injection protocols. AI-based technology addresses behavioral and emotional factors. Youths who forget auto-injectors or therapeutics can be flagged by usage data. Incorporation of AI-driven technology into autoinjectors holds significant strength for improving their primary functions, specifically addressing challenges identified via PMS, like injuries, malfunctions, device and patient challenges.
| Table | Scope |
| Market Size in 2026 | USD 4.42 Billion |
| Projected Market Size in 2035 | USD 12.05 Billion |
| CAGR (2026 - 2035) | 11.8% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Type, By Route of Administration, By Therapy Area, By Technology, By End User, By Distribution Channel, By Region |
| Top Key Players | Eli Lilly and Company, Amgen, Ypsomed, SHL Medical, AbbVie |
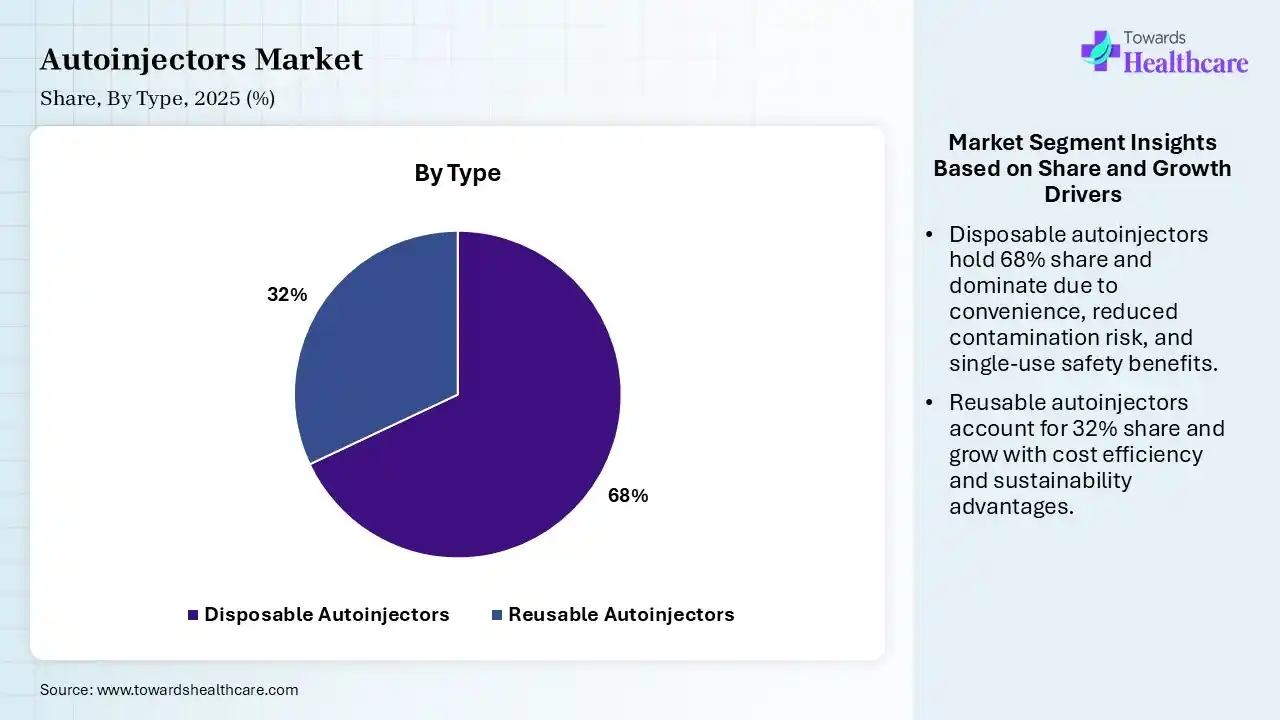
| Segment | Share 2025 (%) |
| Disposable Autoinjectors | 68% |
| Reusable Autoinjectors | 32% |
The Disposable Autoinjectors Segment Led the Autoinjectors Market in 2025
The disposable autoinjectors segment contributed the largest market share of 68% in 2025, as disposable autoinjectors are dense, pre-filled products designed to deliver a particular dose of medication rapidly and safely. Most models involve automated needles, which are hidden before and after injection to prevent needle-stick wounds and lower anxiety.
The reusable autoinjectors segment held a significant share of 32% in the market, and is expected to grow at the fastest CAGR during the forecast period, as it offers important advantages, involving enhanced environmental sustainability by lowering plastic waste, lower long-term expenses for patients using chronic medication, and better patient compliance via digital and connected features.
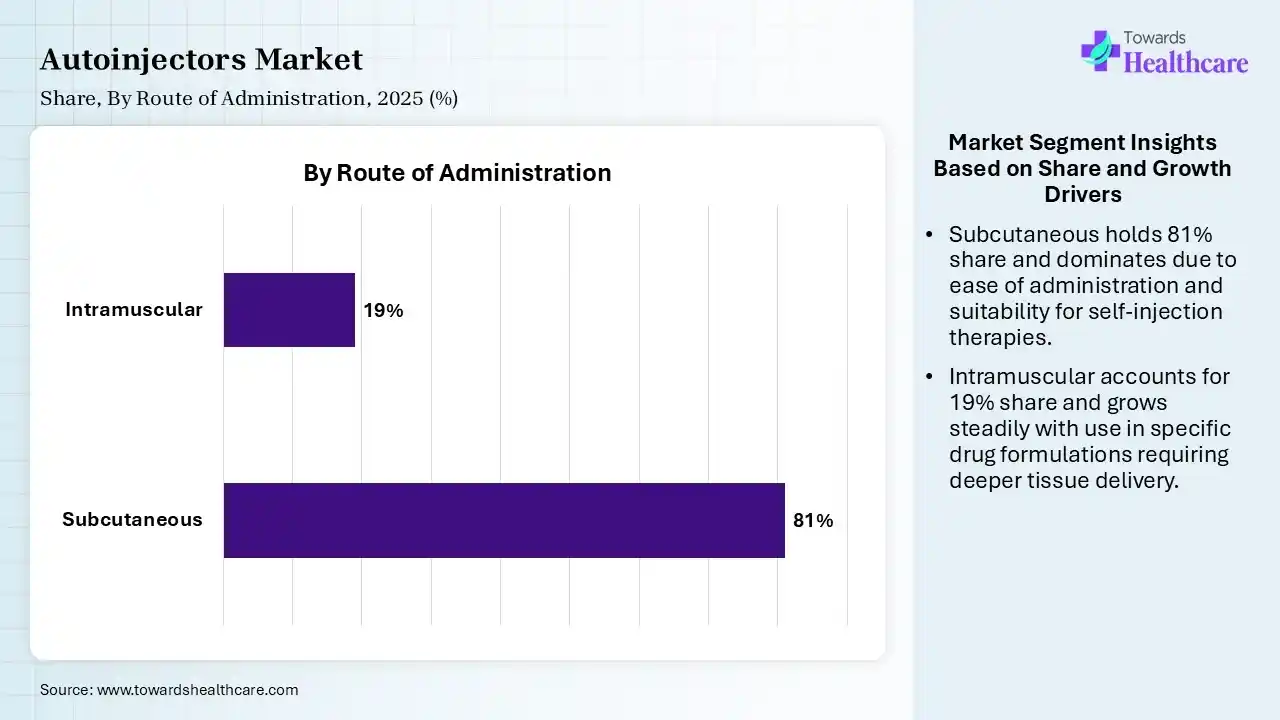
| Segment | Share 2025 (%) |
| Subcutaneous | 81% |
| Intramuscular | 19% |
Subcutaneous Segment Led the Autoinjectors Market in 2025
The subcutaneous segment contributed the largest market share of 81%, and is expected to grow at the fastest CAGR during the forecast period. Subcutaneous administration enables therapeutics to be self-administered by individuals or healthcare providers using a variety of drug delivery technologies, involving advanced subcutaneous drug delivery devices. SC administration eases patient self-administration in home or outpatient healthcare environments.
The intramuscular segment held a significant share of 19% of the market, as intramuscular injection (IM) involves injecting medications into the depth of specifically selected muscles. The bulky muscles have good vascularity, and as the injected drug rapidly reaches the systemic flow and thereafter into the particular area of action, bypassing the first-pass metabolism.
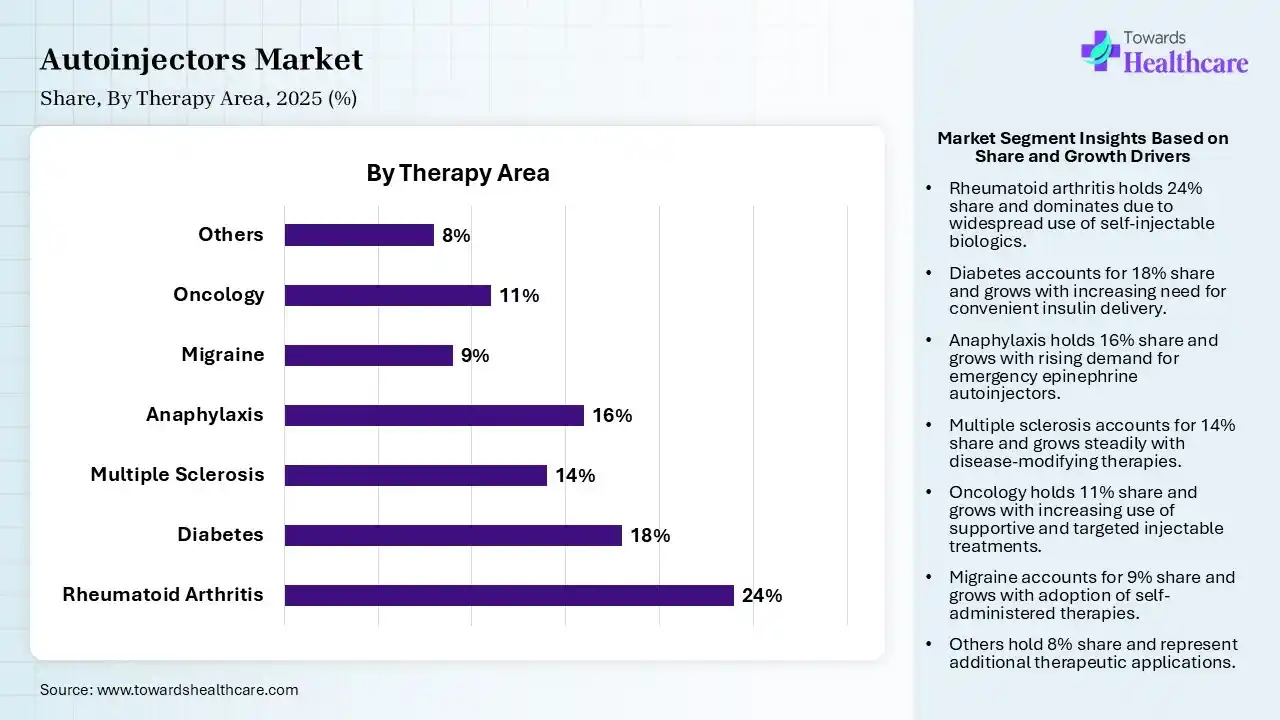
| Segment | Share 2025 (%) |
| Rheumatoid Arthritis | 24% |
| Diabetes | 18% |
| Multiple Sclerosis | 14% |
| Anaphylaxis | 16% |
| Migraine | 9% |
| Oncology | 11% |
| Others | 8% |
Rheumatoid Arthritis Diseases Segment Led the Autoinjectors Market in 2025
The rheumatoid arthritis disease segment contributed the largest market share of 24%, as etanercept is efficient in the management of rheumatoid arthritis (RA) and self-administered through an auto-injector. These autoinjectors offer several advantages, including a simplified drug administration process, safer injection, lower challenges of contamination, and reduced maintenance.
The diabetes segment held a significant share of 18% in the market, as autoinjectors lower the complexity and discomfort of injections, allowing patients to administer rugs with reduced effort. Autoinjectors are specifically well-suited for SC injections because of their less invasive nature, consistent delivery rates, and precise depth control.
The multiple sclerosis segment held a significant share of 14% in the autoinjectors market, as autoinjectors are well-established in subsidiary multiple sclerosis (MS) therapy. Autoinjectors lower the complexity and discomfort of injections, allowing patients to administer medications with minimal effort.
The anaphylaxis segment held a significant share of 16% in the market, and is expected to grow at the fastest CAGR during the forecast period. Autoinjectors are related to higher treatment adherence, with research showing that patients using these tools. This autoinjection prevents the progression of anaphylaxis and thereby reduces the need for hospitalization and the challenges of fatality.
The oncology segment held a significant share of 11% in the market, as autoinjectors deliver specific and consistent doses, lowering the human error related to traditional syringe-driven methods. Autoinjectors in oncology evolved as a patient-driven technology that shifts care from hospitals to the home, addressing the increasing requirement for convenient, self-administered subcutaneous injections of biologics.
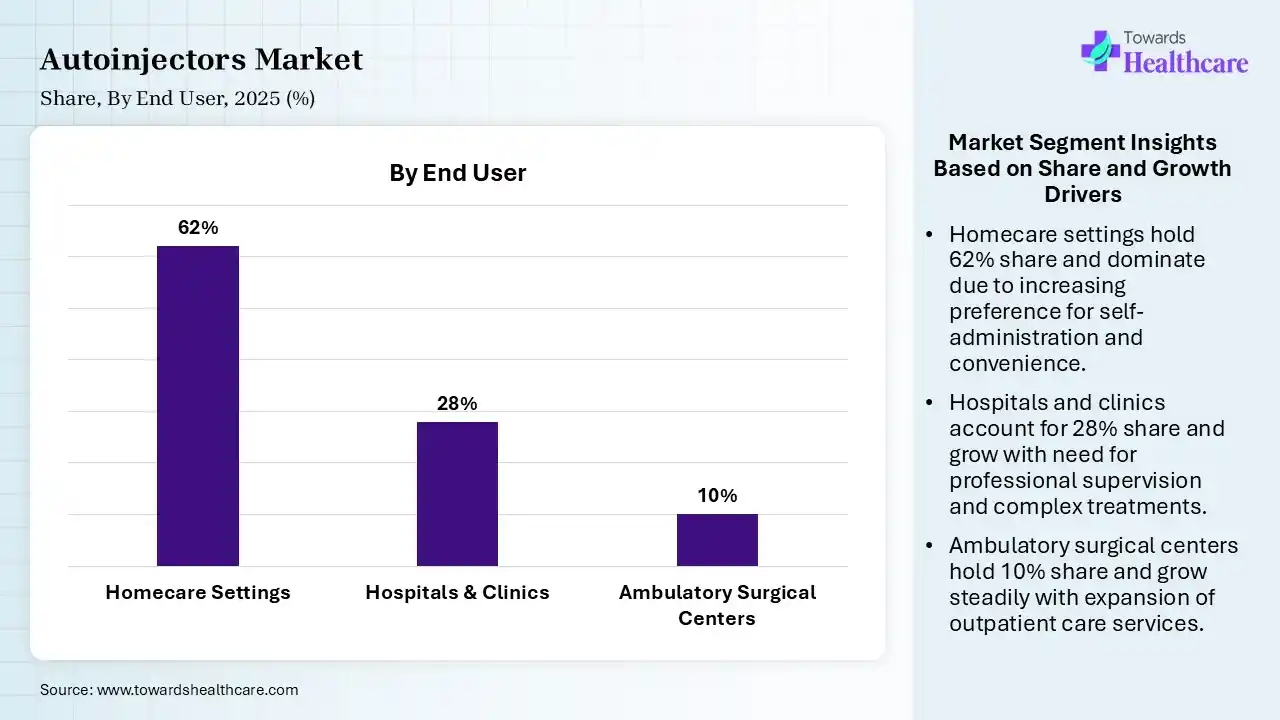
| Segment | Share 2025 (%) |
| Homecare Settings | 62% |
| Hospitals & Clinics | 28% |
| Ambulatory Surgical Centers | 10% |
Homecare Settings Segment Led the Autoinjectors Market in 2025
The homecare settings segment contributed the largest market share of 62%, and is expected to grow at the fastest CAGR during the forecast period, as home care decreases expenses, improves health outcomes, and lowers hospital stays. Home care is focused on preserving the individual's individuality. Home-driven care supports reducing hospital readmissions and enhancing recovery results. By offering services such as medication management and continuing monitoring.
The hospitals & clinics segment held a significant share of 28% in the market, as hospitals and clinics offer significant, accessible healthcare via specialized healthcare staff, advanced technology, and 24/7 emergency care. They offer comprehensive services ranging from preventive check-ups and diagnostic tests to complex surgeries.
The ambulatory surgical centers segment held a significant share of 10% in the market, as ASCs help to cut costs and reduce the out-of-pocket expense for patients. ASCs help to cut costs and reduce the out-of-pocket costs for patients. It is estimated that ASCs can cost 45-60% less than hospital stays. ASCs have better control over the development of surgeries.
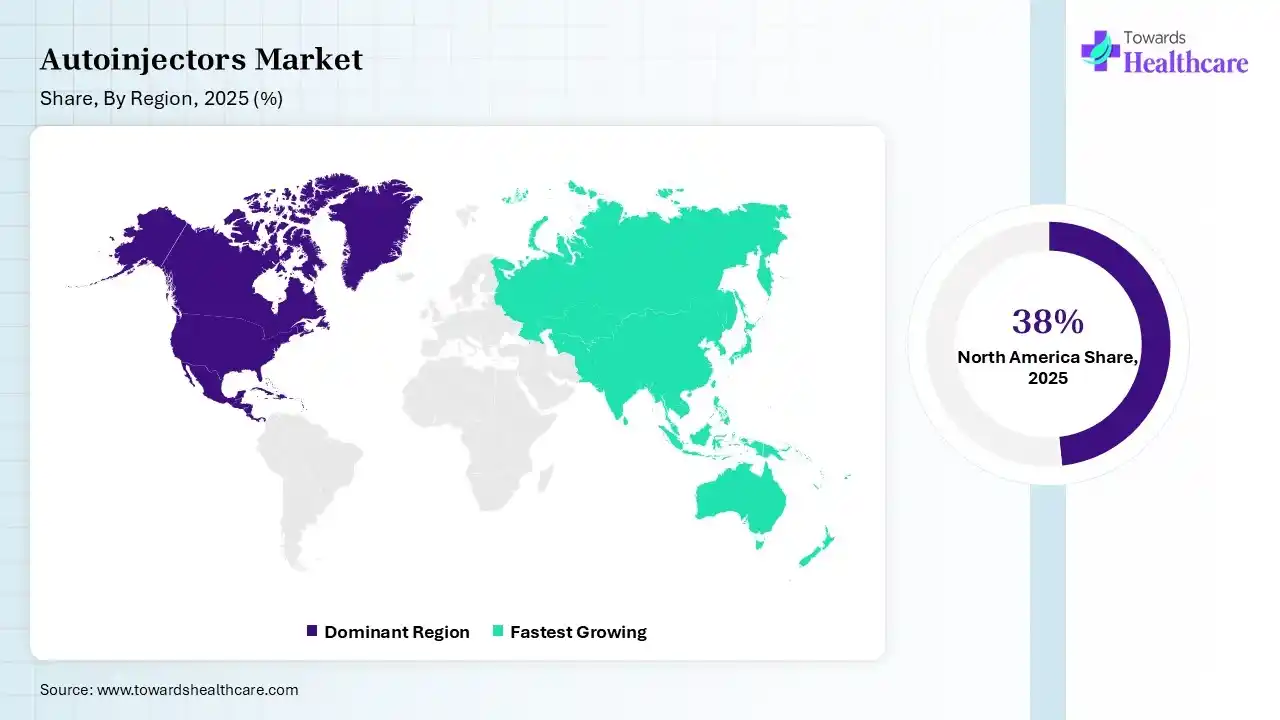
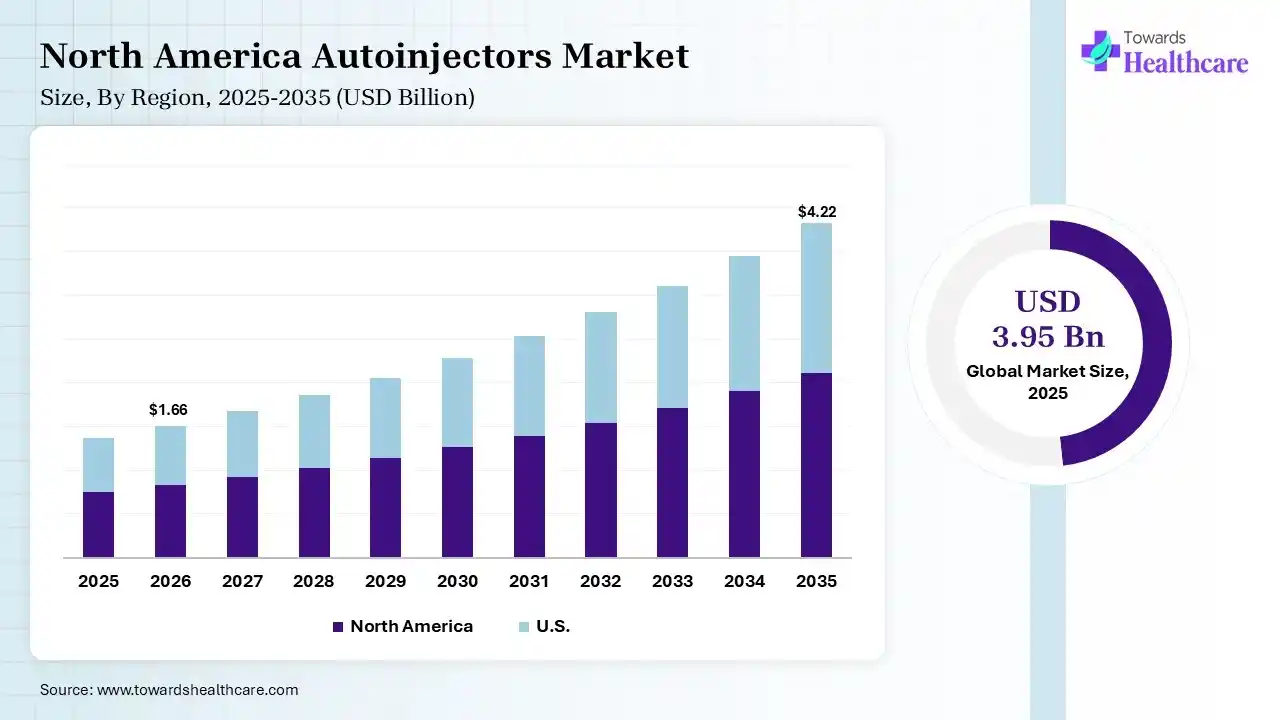
In 2025, North America dominated the autoinjectors market with a share of 38% in 2025, due to increasing chronic conditions like cardiac disease, cancer, stroke, and diabetes, which are expensive and major causes of death and disability in the US. Smart technologies are expected to radically alter care, lowering trips to the hospital and offering greater opportunities to live longer, improved lives at home. The FDA facilitates rapid approvals for novel medical devices, increasing innovation and faster entry for novel, advanced autoinjectors, which contributes to the growth of the market.
For Instance,
U.S. Market Trends
In the U.S., there is a strong preference for home-driven care over clinical visits, making user-friendly, disposable, and connected autoinjectors vital. The U.S. features a high concentration of pharmaceutical organizations investing in biologics and biosimilars, which need advanced, patent-protected, prefilled delivery tools.
Asia Pacific held 23% share of the market, and is expected to have the fastest CAGR during the forecast period, due to a massive, increasing patient population suffering from chronic conditions such as diabetes and rheumatoid arthritis, joined with growing healthcare expenditure and enhanced infrastructure. The region is an increasing hub for biologics and biosimilars in oncology and immunology, which drives the growth of the market.
India Market Trends
India is significantly evolving into a worldwide hub for generic injectable drugs, driven by growing cases of illnesses, affordable production, and solid pharmaceutical exports. The healthcare manufacturing is encountering a significant increase due to the heightened advancement and generation of generic injectables. The growing need for the ease of drug administration, which increases the demand for autoinjectors.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Eli Lilly and Company | United States | In February 2026, Eli Lilly and Company announced the U.S. Food and Drug Administration (FDA) approved a label expansion for Zepbound to include the four-dose single-patient use KwikPen that delivers a full month of treatment in one tool. |
| Amgen | United States | In October 2025, Amgen announced the launch of AmgenNow, a novel direct-to-patient program starting with Repatha. |
| Ypsomed | United States | In October 2026, Ypsomed launches YpsoLoop, the first autoinjector platform designed for circularity. The two new 1 ml and 2 ml platform versions enable material recovery through a design enabling automated disassembly. |
| SHL Medical | United States | In March 2025, SHL Medical AG, the worldwide leader in advanced drug delivery services, announced a strategic collaboration with Thermo Fisher Scientific Inc., the world leader in serving science. |
| AbbVie | United States | In April 2026, AbbVie submitted a supplemental application to the FDA seeking approval for subcutaneous (SC) risankizumab-rzaa as an induction therapy for adults with moderately to severely active Crohn disease. |
Strengths
Weakness
Opportunities
Threat
By Type
By Route of Administration
By Therapy Area
By Technology
By End User
By Distribution Channel
By Region
May 2026
May 2026
May 2026
May 2026