December 2025

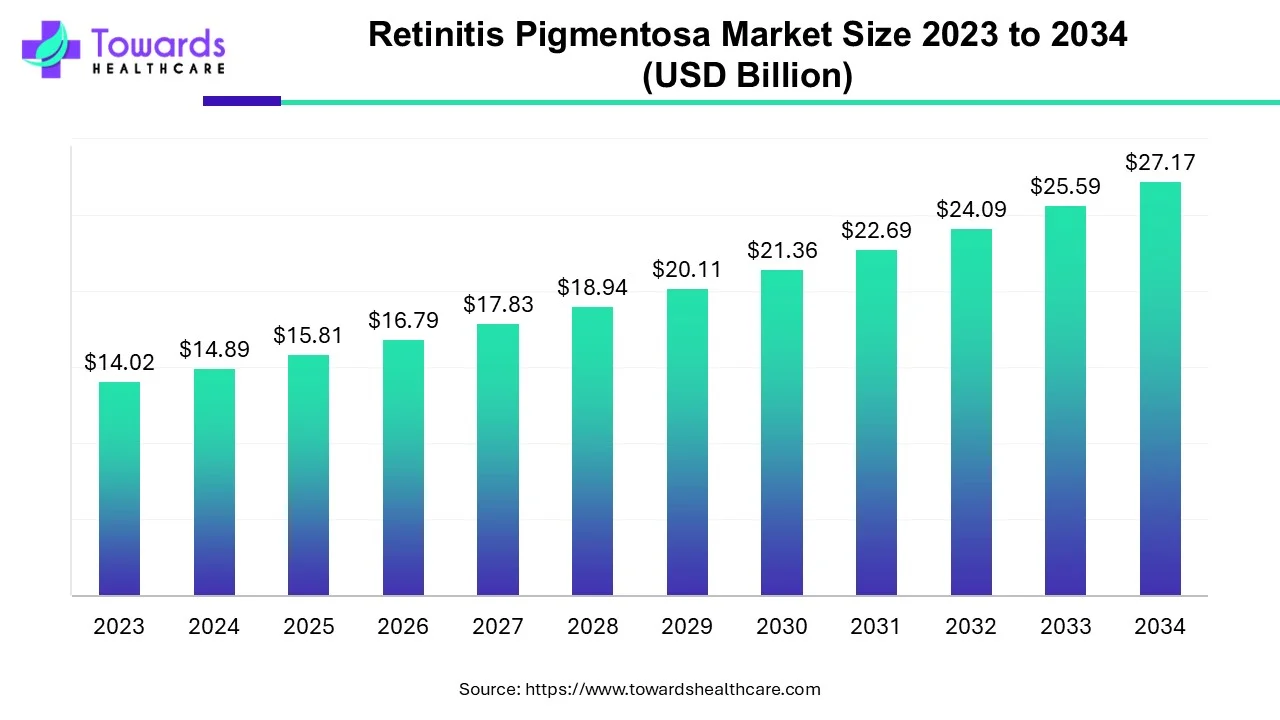
The retinitis pigmentosa market is predicted to expand from USD 15.81 billion in 2025 to USD 27.17 billion by 2034, growing at a CAGR of 6.2% during the forecast period from 2025 to 2034.
In 2022, approximately 82,500 to 110,000 individuals in the United States were affected by retinitis pigmentosa. This underscores the growing demand for innovative approaches to address the condition, such as gene therapy and retinal implants.
Retinitis Pigmentosa comprises genetic eye disorders impacting the retina, the eye's vision center. It results in the deterioration of photoreceptor cells crucial for vision in various light conditions and clarity. As it progresses, individuals may encounter night or low-light vision challenges, reduced peripheral vision, and potentially total blindness.
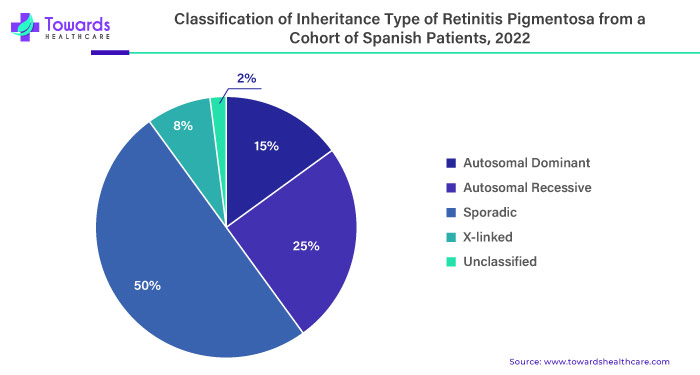
Retinitis Pigmentosa is divided into two main types: non-syndromic and syndromic. Non-syndromic retinitis pigmentosa, which makes up 70-80% of cases, doesn't involve other systemic abnormalities. Syndromic retinitis pigmentosa, accounting for 20-30% of cases, is associated with additional non-ocular syndromes and systemic diseases. Non-syndromic retinitis pigmentosa is linked to mutations in 80 different genes, with around 20% being autosomal recessive, 10-20% autosomal dominant (AD), and 10% X-linked recessive.
For instance,
The importance of understanding and addressing retinitis pigmentosa lies in its impact on individuals' quality of life. Vision loss can significantly affect a person's independence, mobility, and overall well-being. Additionally, retinitis pigmentosa is often hereditary, meaning it can affect multiple generations within a family. Advancements in research, diagnosis, and treatment options for retinitis pigmentosa are essential for improving outcomes and providing hope for affected individuals and their families.
In 2023, the CDC revealed that around 12 million Americans aged 40 and above experience vision impairment, including 1 million who are blind, 3 million with correctable vision issues, and 8 million due to untreated refractive errors. As the global population ages and awareness of genetic diseases rises, there's a growing demand for retinitis pigmentosa treatments and technologies. Precision medicine and gene therapy are gaining traction for addressing genetic disorders like retinitis pigmentosa.
Furthermore, advancements in gene editing, stem cell therapy, and retinal implants offer promising avenues for managing retinitis pigmentosa and enhancing visual function. Increased investment in research and development by biotech and pharmaceutical firms, academic institutions, and government bodies bolsters the retinitis pigmentosa treatment market. Moreover, regulatory agencies are streamlining approval processes for innovative therapies, potentially expediting the availability of new retinitis pigmentosa treatments. The market for retinitis pigmentosa treatments and technologies is expected to grow steadily as scientific understanding improves, new therapeutic approaches emerge, and the demand for solutions to address vision loss continues to rise.
Early detection and diagnosis of retinitis pigmentosa are super important because they help catch the condition early, allowing doctors to start the right treatments sooner. Techniques like genetic testing and eye exams can spot Retinitis Pigmentosa before it worsens, giving people a better chance at managing it. Vision rehab is also crucial. It helps folks with retinitis pigmentosa learn new ways to navigate the world with their changing vision, using tools like special glasses or learning mobility skills.
Genetic counseling is essential for families affected by retinitis pigmentosa. It helps them understand how the condition can be passed down and make intelligent choices about family planning. Research is ongoing to find new treatments for retinitis pigmentosa, like gene therapy or stem cell therapy, which could slow or even reverse the damage. Additionally, psychosocial support is crucial for people living with retinitis pigmentosa. It offers them emotional support, counseling, and connections with others who understand what they're going through, helping them cope better with the challenges of vision loss.
In 2022, the National Institute of Health estimated that retinitis pigmentosa is the most common inherited retinal disorder, affecting over 1.5 million people worldwide. The rise in retinitis pigmentosa cases globally is mainly due to two reasons: more people getting older and an increase in genetic disorders. People living longer are more likely to develop retinitis pigmentosa, often appearing later in life. Since retinitis pigmentosa is mainly passed down through families, better genetic testing and awareness have identified more cases. Also, when relatives intermarry, it can add to the number of retinitis pigmentosa cases. With more people being diagnosed with retinitis pigmentosa, there's a greater need for effective treatments. This growing need opens up opportunities for research to find new therapies and improve existing ones. It's essential for healthcare and research to keep working on understanding retinitis pigmentosa and finding better ways to manage it as the number of affected individuals continues to grow.
Gene therapy is used in the treatment approach for Retinitis Pigmentosa, aiming to address the underlying genetic mutations responsible for the condition. Retinitis Pigmentosa encompasses diverse inherited retinal disorders, many caused by mutations in specific genes essential for normal retinal function. Gene therapy seeks to correct the genetic defects underlying RP and restore vision by delivering functional copies of these genes into retinal cells.
Recent News:
Gene therapy for Retinitis Pigmentosa typically involves using viral vectors or other delivery systems to transport therapeutic genes into the retina. Viral vectors, such as adeno-associated viruses (AAVs), are often employed because they efficiently deliver genes to target cells without causing harmful immune responses. Once introduced into the retina, the therapeutic genes integrate into the retinal cells and produce functional proteins necessary for proper visual function.
Clinical trials investigating gene therapy for retinitis pigmentosa have shown promising results, with some patients experiencing improved vision following treatment. These improvements may include slowed disease progression, preservation of remaining vision, or, in some cases, partial restoration of lost vision. Gene therapy holds particular promise as a targeted treatment approach for individuals with specific genetic mutations associated with retinitis pigmentosa.
For instance,
As gene therapy techniques continue to be refined and optimized, there is growing optimism regarding their potential to slow or halt the progression of retinitis pigmentosa in affected individuals. Ongoing research efforts aim to improve gene therapy's safety, efficacy, and delivery methods for retinitis pigmentosa, further enhancing its therapeutic potential.
Due to the promising results observed in clinical trials and the increasing understanding of the genetic basis of retinitis pigmentosa, the market for gene therapy in retinitis pigmentosa treatment has experienced significant growth. Pharmaceutical companies, biotechnology firms, and research institutions are investing resources into developing and commercializing gene therapy products for retinitis pigmentosa, driving market expansion. Additionally, regulatory agencies have shown interest in expediting the approval process for gene therapy treatments for rare genetic disorders like retinitis pigmentosa, further contributing to market growth.
Gene therapy represents a groundbreaking approach to treating retinitis pigmentosa by targeting the underlying genetic defects responsible for the condition. As gene therapy techniques advance, they offer hope for improving outcomes and quality of life for individuals affected by retinitis pigmentosa, leading to increased market demand and investment in retinitis pigmentosa treatment options.
Retinal implants, also known as artificial retinas or bionic eyes, represent an innovative technology designed to restore vision in individuals with severe vision loss, such as those affected by Retinitis Pigmentosa. These innovative devices electrically stimulate the remaining retinal cells to generate visual perceptions, bypassing the retina's damaged or non-functioning photoreceptor cells.
For instance,
The essential components of retinal implants typically include an electrode array implanted on the retina and an external camera system. The camera system captures visual information from the surrounding environment and processes it into electrical signals transmitted to the electrode array. The electrode array stimulates the remaining retinal cells, such as ganglion cells, which transmit visual information to the brain, thus generating visual perceptions.
While current retinal implants have demonstrated the ability to restore some degree of vision in individuals with severe vision loss, they still have limitations regarding visual acuity and resolution. Ongoing research efforts aim to overcome these limitations and enhance the performance and functionality of retinal implants. One area of focus in retinal prosthesis research is improving electrode design to achieve more precise and targeted stimulation of retinal cells. Researchers aim to enhance electrical stimulation's specificity and effectiveness by optimizing electrode materials, shapes, and configurations, thereby improving user visual outcomes.
Additionally, advancements in surgical techniques are crucial in retinal implants' successful implantation and functioning. Minimally invasive surgical approaches, precise electrode array placement, and tissue integration improvements are essential for maximizing retinal implant device's long-term efficacy and safety.
Ongoing research aims to expand the applicability of retinal prostheses to a broader range of retinitis pigmentosa patients, including those with varying degrees of retinal degeneration. By addressing current limitations and improving performance and functionality, retinal implants hold tremendous promise for significantly improving the quality of life for individuals with severe vision loss due to conditions like retinitis pigmentosa.
Developing new therapies for retinitis pigmentosa, such as gene therapy and stem cell therapy, is multifaceted and costly. The process begins with extensive primary research to uncover the underlying causes of retinitis pigmentosa and identify potential therapeutic targets. This phase involves conducting experiments in laboratory settings, which require specialized equipment, reagents, and expertise. Following this, preclinical studies evaluate potential therapies' safety, efficacy, and pharmacokinetics using animal retinitis pigmentosa models. These studies are crucial for understanding how the treatments may work before moving on to human trials.
In 2023, the World Health Organization (WHO) stated that vision problems create a significant financial strain worldwide, with the yearly global productivity cost estimated at USD 411 billion. Rising treatment expenses present challenges in the Retinitis Pigmentosa market. Clinical trials represent the final testing phase, involving multiple stages (Phase I to Phase III) to assess the therapies' safety, dosage, and efficacy in human participants. Clinical trials are highly regulated, requiring careful planning, coordination, and compliance with stringent regulatory standards set by agencies such as the Food and Drug Administration (FDA). Meeting these standards entails additional costs and complexities, including securing regulatory approvals, recruiting and retaining participants, and managing trial data.
As therapies progress through clinical development, efforts are made to scale up production for potential commercialization. This requires establishing robust manufacturing processes and facilities to ensure product consistency, quality, and compliance with Good Manufacturing Practice (GMP) standards. Additionally, protecting intellectual property through patents is essential for securing market exclusivity and attracting investment in retinitis pigmentosa therapies. However, obtaining and maintaining patents can be expensive and time-consuming.
Small biotechnology companies and academic researchers often need help securing the necessary funding to support the development process. They rely on various funding sources, including government grants, private investments, collaborations with larger companies, and philanthropic support, to advance retinitis pigmentosa treatments from the laboratory to clinical use. Despite the obstacles, continued investment and cooperation are essential for driving progress in developing effective therapies for RP and improving outcomes for individuals affected by this debilitating condition.
North America, particularly the United States, is a significant hub for retinitis pigmentosa research, development, and market activity. The region boasts leading academic institutions, research centers, and biotechnology companies focused on ophthalmic research and innovative therapies for retinitis pigmentosa. Additionally, the U.S. Food and Drug Administration (FDA) is central in regulating RP treatments and approving new therapies for commercialization.
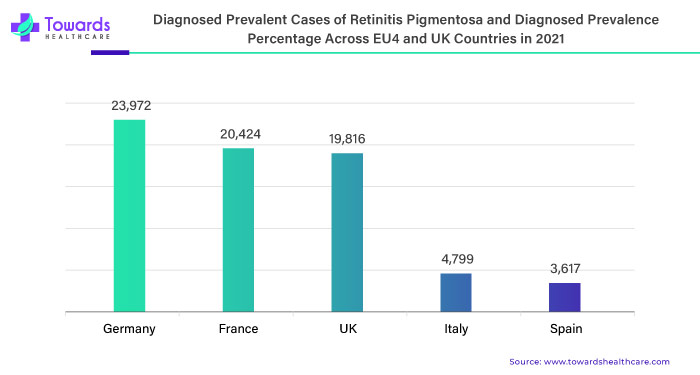
The geographic landscape of the retinitis pigmentosa market in Europe encompasses various regions with differing prevalence levels, access to healthcare, and regulatory frameworks. Countries such as the United Kingdom, Germany, France, Italy, and Spain are prominent markets for retinitis pigmentosa treatment and research in Europe due to their well-established healthcare systems and significant patient populations.
The Asia-Pacific region is emerging as a growing market for retinitis pigmentosa treatments, fueled by advancements in healthcare infrastructure, increasing awareness of genetic disorders, and rising healthcare expenditure. Countries like Japan, China, and South Korea invest in ophthalmic research and development while serving as essential markets for retinitis pigmentosa therapies.
The competitive landscape of the Retinitis Pigmentosa market encompasses a range of stakeholders, including pharmaceutical companies, biotechnology firms, academic institutions, research organizations, and healthcare providers. These entities are engaged in various activities, such as research and development of novel therapies, manufacturing and commercialization of existing treatments, and provision of healthcare services to retinitis pigmentosa patients. Government agencies, such as the National Institutes of Health (NIH) in the United States and the European Commission's Horizon 2020 program, provide funding and support for research and development initiatives for retinitis pigmentosa. These agencies play a crucial role in fostering innovation, facilitating collaborations, and advancing therapeutic discoveries in the field of ophthalmology.

By Disease Type
By Treatment Type
By Geography
December 2025
November 2025
November 2025
February 2026
