May 2026

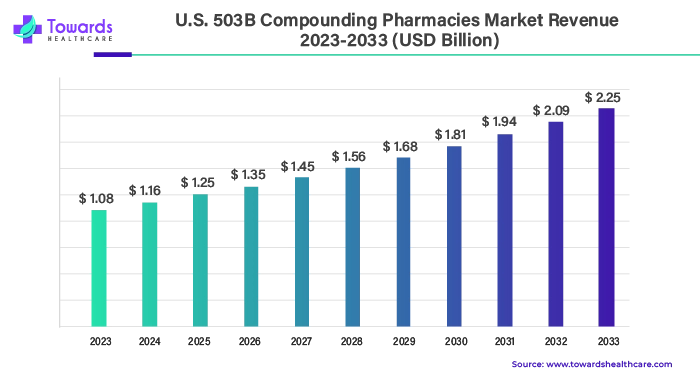
The U.S. 503B compounding pharmacies market size is calculated at US$ 1.16 billion in 2024, grew to US$ 1.25 billion in 2025, and is projected to reach around US$ 2.42 billion by 2034. The market is expanding at a CAGR of 7.63% between 2025 and 2034.
503B compounding pharmacies are FDA-regulated facilities permitted to produce sterile medications in bulk, unlike traditional 503A pharmacies that can only compound drugs based on individual prescriptions. These pharmacies adhere to Current Good Manufacturing Practices (CGMP) and are routinely inspected by the FDA, ensuring the highest quality and safety standards.
But what sets them apart is their ability to produce and stock ready-to-use, sterile injectables, medications that are vital for emergency rooms, intensive care units, and high-demand clinical environments.
Expanding industries: To accelerate the development of compounded drugs, the industries are increasingly investing in the 503B compounding pharmacies. Moreover, new acquisitions are also contributing to the same, leading to the expansion of the industries. Moreover, new guidelines are also being provided to support the development of the drugs in these pharmacies.
For instance,
With a surge in infectious diseases, chronic conditions, and emergency care needs, the use of sterile injectables has witnessed a major spike. These drugs are critical in ICUs, ERs, and surgical centers, especially for:
According to healthcare data, the global demand for sterile injectable drugs is expected to grow by over 7% annually over the next decade, giving 503B pharmacies a huge opportunity to scale.
Artificial Intelligence is reshaping the pharmaceutical world and 503B pharmacies are no exception. AI is now used to:
In fact, facilities leveraging AI in compounding have reported up to a 40% reduction in errors and significantly faster turnaround times.
Global drug shortages are at an all-time high, with over 300 medications listed as in short supply in the U.S. alone (FDA, 2025). Hospitals are increasingly relying on 503B compounding pharmacies to fill this critical gap.
By producing essential sterile medications in advance and at scale, 503B facilities are helping healthcare providers avoid treatment delays, ultimately improving patient outcomes.
In January 2025, the FDA released new guidance supporting the use of bulk drug substances for compounding under both 503A and 503B. This regulatory update not only clarifies the rules but also empowers compounding pharmacies to innovate and expand their drug offerings legally and safely.
This kind of regulatory transparency boosts confidence among both pharmaceutical companies and healthcare systems, ensuring faster adoption and investment.
Like any highly regulated industry, 503B compounding comes with its share of challenges. Regulatory hurdles, including stringent CGMP requirements and unannounced FDA inspections, can slow development. However, companies that invest in automation, AI, and robust quality systems are finding ways to not just survive, but thrive.
North America is the undisputed leader in the 503B compounding pharmacy space. With a highly advanced healthcare infrastructure, the region depends heavily on these outsourcing facilities to meet the rising demand for ready-to-use, sterile injectables in high-pressure environments like ICUs, emergency rooms, and surgical centers.
Hospitals and clinics across the U.S. are increasingly relying on 503B pharmacies for cancer drugs, anesthesia medications, and antibiotics. The legal framework supporting large-scale compounding allows for cost-effective, standardized, and safer drug delivery in clinical settings.
In Canada, sterile compounding is gaining traction as healthcare facilities look for safer, more consistent drug supply chains. Backed by federal and provincial funding, new guidelines from regulatory bodies are encouraging pharmacies to upgrade compounding capabilities.
SCA Pharma
SCA Pharma specializes in customized sterile injectable solutions, offering a wide range of compounded medications for hospitals, health systems, and surgery centers across the U.S.
Athenex, Inc.
Athenex provides high-quality, sterile injectable drugs through its 503B outsourcing facility, focusing on oncology and critical care medications.
Nephron Pharmaceuticals Corporation
Nephron is known for its large-scale production of sterile injectables and respiratory medications, supplying hospitals and healthcare providers nationwide.
Wells Pharma of Houston LLC
Wells Pharma offers 503B sterile compounded products with a focus on regulatory compliance, safety, and fast turnaround for emergency and surgical care.
Fagron Compounding Pharmacies
Fagron is a global leader in pharmaceutical compounding, delivering innovative sterile and non-sterile solutions tailored to individualized patient and provider needs.
Olympia Pharmacy
Olympia Pharmacy provides a broad portfolio of 503B compounded injectables, with specializations in wellness, hormone therapy, and aesthetic medicine.
In July 2025, after receiving the 2025 Pharmacy Compounding Excellence Award, the chief pharmacy officer of BayCare Health System, Mike Magee, stated that, on behalf of BayCare Health System, he was pleased to receive this award. They were able to build one of four health system FDA-approved 503B compounding facilities in the U.S., under the expertise and leadership of their colleague Ken Jozefczyk. With this center, they were able to offer quality treatments to the patients without any drug shortages across the Tampa Bay community.
May 2026
May 2026
May 2026
May 2026