March 2026

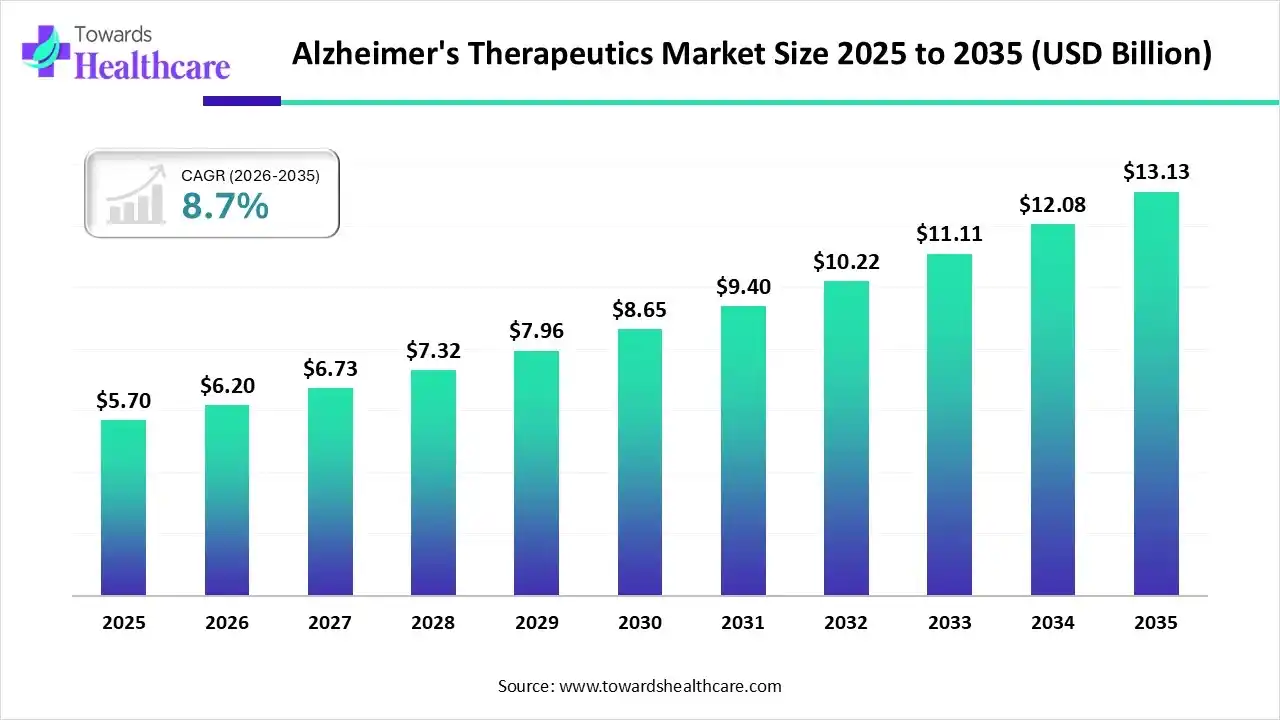
The global alzheimer’s therapeutics market size is projected to reach USD 13.13 billion by 2035, growing from USD 5.7 billion in 2025, at a CAGR of 8.7% during the forecast period from 2026 to 2035. The rising prevalence of Alzheimer’s disease, growing research and development activities, and demand for personalized care drive the market.
| Key Elements | Scope |
| Market Size in 2026 | USD 6.2 Billion |
| Projected Market Size in 2035 | USD 13.13 Billion |
| CAGR (2026 - 2035) | 8.7% |
| Leading Region | North America |
| Market Segmentation | By Product Type, By End User, By Geography |
| Top Key Players | Biogen Inc., Eli Lilly and Company, Eisai Co., Ltd, F. Hoffmann-La Roche Ltd., GlaxoSmithKline plc, Johnson & Johnson Services Inc., Merck & Co., Inc., Novartis AG, Pfizer Inc, AbbVie Inc. |
The National Center for Biotechnology Information reported that in 2021, around 6.2 million Americans aged 65 and older had Alzheimer's disease. By 2060, this number is expected to double to about 13.8 million nearly. Women are more likely to get Alzheimer's because they tend to live longer. Alzheimer's is becoming the top cause of death among neurodegenerative diseases and can lead to physical disability, needing prompt treatment.
Alzheimer's disease is a type of dementia that affects memory, thinking, and behavior. It's caused by changes in the brain that lead to the death of brain cells. These changes can make it hard for people with Alzheimer's to remember things, communicate, and take care of themselves.
The exact cause of Alzheimer's disease isn't fully understood, but scientists believe it's a combination of genetic, environmental, and lifestyle factors. As people age, their risk of developing Alzheimer's disease increases, but it can also affect younger people.
It's essential to treat and manage Alzheimer's disease because it can significantly affect a person's quality of life and ability to function independently. There's currently no cure for Alzheimer's disease, but treatment options are available to help manage symptoms and slow down the progression of the disease. This can include medications to improve memory and thinking and therapies to help with communication and daily activities.
Additionally, proper management of Alzheimer's disease can help improve the quality of life for both the person with the disease and their caregivers. This may involve creating a safe and supportive environment, providing assistance with daily tasks, and offering emotional support and resources for caregivers. Early diagnosis and intervention are crucial to helping people with Alzheimer's disease live as independently and comfortably as possible for as long as possible.
Artificial intelligence (AI) plays a crucial role in developing novel therapeutics for the treatment of Alzheimer’s disease (AD). AI can assess the patient’s brain conditions and predict the severity of the disease. It can also evaluate biomarkers and other targets involved in AD. This helps researchers to develop novel therapeutics. AI can streamline the entire research process, enhancing precision and accuracy. AI-driven approaches can develop personalized treatments based on patients’ conditions. AI can also predict the interactions of drugs and targets within the body. Several researchers also focus on the drug repurposing of existing drugs for novel indications. AI can revolutionize drug repurposing approaches by screening lakhs of drugs against AD targets.
As people get older, they become more likely to develop Alzheimer's disease. This is because aging is one of this condition's most significant risk factors. As the population around the world ages, more and more people are entering the age range where they are at higher risk for Alzheimer's. This means that more people may develop the disease and need treatment. As a result, there's a greater demand for Alzheimer's therapeutics – medications and other medicines that can help manage the symptoms and slow down the progression of the disease.
Imagine if you have a big group of people, and as they get older, more of them start to have trouble with their memory and thinking. This is like what happens with Alzheimer's disease in the elderly population. Because there are more older people, there are also more people who might need help with Alzheimer's.
For instance,
This increase in demand for Alzheimer's therapeutics is a big reason researchers, doctors, and pharmaceutical companies are working hard to develop new treatments. They want to find ways to help people with Alzheimer's live better lives for longer, even as they get older. So, understanding why aging increases the risk of Alzheimer's and finding ways to treat it is essential for helping older adults stay healthy and independent.
Researchers all around the world are working hard to figure out why Alzheimer's disease happens and how we can treat it better. Alzheimer's is a complex condition that affects the brain, and there's still a lot we don't understand about it. That's why scientists are doing ongoing research – they want to uncover the secrets of Alzheimer's and find new ways to help people with it.
One important part of this research is trying to understand what goes wrong in the brain of someone with Alzheimer's. Scientists are studying things like the buildup of specific proteins in the brain and how they might affect brain cells. They're also looking at how brain cells communicate with each other and how this communication might be disrupted in Alzheimer's.
Another focus of research is finding new treatments for Alzheimer's. Scientists are always looking for ways to develop better medications or other therapies that can help slow down the progression of the disease or improve symptoms. This might involve testing new drugs in the lab or in clinical trials with people who have Alzheimer's.
One exciting area of research is the search for biomarkers. Biomarkers are things in the body that tell us if someone has a particular disease or how severe it is. For Alzheimer's, researchers are looking for biomarkers that can help with early diagnosis or tracking the progression of the disease over time. This could lead to better treatment options and more personalized care for people with Alzheimer's.
| Sr. No. | Approved Date | Drug Name | Indication |
| 1. | In March 2022 | Adlarity (donepezil hydrochloride) | It's used to treat severe Alzheimer's disease and is applied once a week on the skin. This patch provides a steady dose of the medication. Adlarity is the fourth drug of its kind to be approved and released in the U.S. The Alzheimer's treatment market is categorized based on different types of drugs, including cholinesterase inhibitors like Adlarity, NMDA receptor antagonists, combination drugs, and drugs still in development. |
| 2. | In June 2021 | Aduhelm (aducanumab) | In Alzheimer's disease research, a major focus is on preventing the buildup of amyloid beta (Aβ) and the formation of Aβ plaques in the brain. |
| 3. | In January 2023 | Leqembi (lecanemab) | Leqembi (lecanemab) is a medication used to treat Alzheimer's disease. It works by targeting and reducing the buildup of amyloid beta plaques in the brain, which are associated with Alzheimer's progression. By addressing this underlying cause, Leqembi aims to potentially slow down the decline in cognitive function experienced by Alzheimer's patients. |
This research is vital because it helps us learn more about Alzheimer's and how to fight it. Every discovery brings us closer to finding better treatments and, hopefully, a cure for this devastating disease. So, by supporting research efforts, we're giving hope to millions of people worldwide affected by Alzheimer's.
Hospitals are essential for helping people with Alzheimer's disease. They have special teams of doctors and nurses who know much about Alzheimer's and how to treat it. In hospitals, patients can get checked with fancy machines like MRIs and PET scans, which help doctors see what's happening in their brains. This helps with diagnosing Alzheimer's early, which is essential for getting the proper treatment and making sure patients do as well as possible.
At hospitals, patients can also get help from lots of different experts, like brain doctors (neurologists), old-age doctors (geriatricians), and mental health doctors (psychiatrists). These experts all work together to ensure patients get the best care possible. Hospitals also offer treatments for Alzheimer's, like medicines, therapy to help with memory and thinking, and support services for patients and their families.
Some hospitals even research to find new treatments for Alzheimer's. They run studies to test new medicines and therapies to see if they can help patients even more. And for patients who need extra help because their Alzheimer's is evil, hospitals have special units where they can stay and get lots of care and support.
Hospitals are the principal place where people with Alzheimer's go for help. They help diagnose the disease, offer different treatments, research to find new treatments and support patients and their families. As more and more people get Alzheimer's as they get older, hospitals will keep playing a significant role in making sure everyone gets the help they need.
Regulatory hurdles and challenges in clinical trial design can make it difficult for new Alzheimer's treatments to be developed and approved. These challenges are mainly because government agencies like the Food and Drug Administration and the European Medicines Agency have strict rules about how drugs must be tested to ensure they work and are safe for people to use. These rules can slow the development of new medicines and make it more expensive.
When it comes to Alzheimer's, designing clinical trials – which are the tests done to see if a new drug works – can be especially tricky. One challenge is determining what to measure to see if the drug is helping. Since Alzheimer's affects people differently, it's essential to pick the right things to measure that show if the drug is making a difference.
Another challenge is deciding who should be in the clinical trials. Researchers need to pick people who have Alzheimer's, but they also need to make sure they're similar enough to each other so they can get accurate results. This can be tough because Alzheimer's affects people differently, and they may have other health problems, too.
Dealing with these regulatory hurdles and challenges in clinical trial design takes a lot of careful planning and attention to detail. But it's essential to make sure that any new Alzheimer's treatments are safe and work before they're available to the public.
By product, the cholinesterase inhibitors segment held a dominant presence in the market in 2024. Cholinesterase inhibitors inhibit the enzyme cholinesterase responsible for the breakdown of acetylcholine, potentiating the formation of acetylcholine. This leads to cognitive decline and dysregulation of cholinesterase enzyme. Common examples of cholinesterase inhibitors include donepezil, rivastigmine, and pyridostigmine. The majority of AD cases, about 60-70%, are characterized by cognitive decline. Hence, cholinesterase inhibitors are the widely prescribed therapeutics for AD.
By product, the pipeline drugs segment is expected to grow at the fastest rate in the market during the forecast period. The growing research and development activities have led to the development of novel therapeutics for AD. This results in an increasing number of clinical trials. In 2024, the Alzheimer’s disease drug development pipeline contained 127 drugs evaluated in 164 clinical trials. Currently, there are no drugs available on the market that target the root cause of the disease. Hence, the hunt for novel therapeutics continues that not only provides symptomatic relief but also treats the disease from its root cause.
By end-user, the hospital pharmacy segment led the global market in 2024. The increasing number of hospitalizations for regular check-ups facilitates the purchase of medications from a hospital pharmacy. Hospital pharmacies contain different types of medications for all conditions. The presence of skilled professionals and favorable infrastructure augment the segment’s growth. Hospitals are also part of clinical trials, benefitting patients to participate in trials and get access to novel medications before market approval.
By end-user, the e-commerce segment is predicted to witness significant growth in the market over the forecast period. As Alzheimer’s disease is mainly caused in older age populations, the increasing geriatric population boosts the segment’s growth. Patients can order their medicines in the comfort of their homes at any time. Online pharmacies also provide reminders to refill their monthly prescriptions. They offer same-day home delivery and special discounts, attracting more customers.
In North America, lots of people have Alzheimer's disease, so the market for Alzheimer's treatments is big. The region has excellent hospitals and research centers, so Alzheimer's can be diagnosed early, and people can get the newest treatments. The FDA and Health Canada ensure Alzheimer's drugs are safe and work well. North America spends a lot of money on healthcare, and groups that support Alzheimer's patients also help make the market better. Overall, North America is critical for Alzheimer's research and treatment.
In the Asia Pacific region, the Alzheimer's Therapeutics Market is growing because more people are getting older, leading to more cases of Alzheimer's. This means there's a higher demand for treatments. Countries are also improving their healthcare systems, which creates more opportunities for the market to grow. Places like China, Australia, and India, where many people and more money are being spent on healthcare, are significant for the market. Dealing with different rules and regulations in each country can take much work for companies.
For instance,
But, there's a lot of focus on researching and making new treatments that fit the region's needs. The Asia Pacific region has good chances for the Alzheimer's Therapeutics Market to grow and improve at diagnosing, treating, and managing the disease.
Rita Balice-Gordon, CEO of Muna Therapeutics, commented on the collaboration with GSK, saying that the agreement marks a pivotal moment in Muna’s evolution and the broader Alzheimer’s research landscape. Combining GSK’s commitment with the company’s MiND-MAP platform will help to deliver novel insights into brain resilience. The company aims to transform the landscape of drug discovery for neurodegenerative diseases and bring new hope to millions of patients worldwide.

By Product Type
By End User
By Geography
March 2026
March 2026
March 2026
March 2026
