
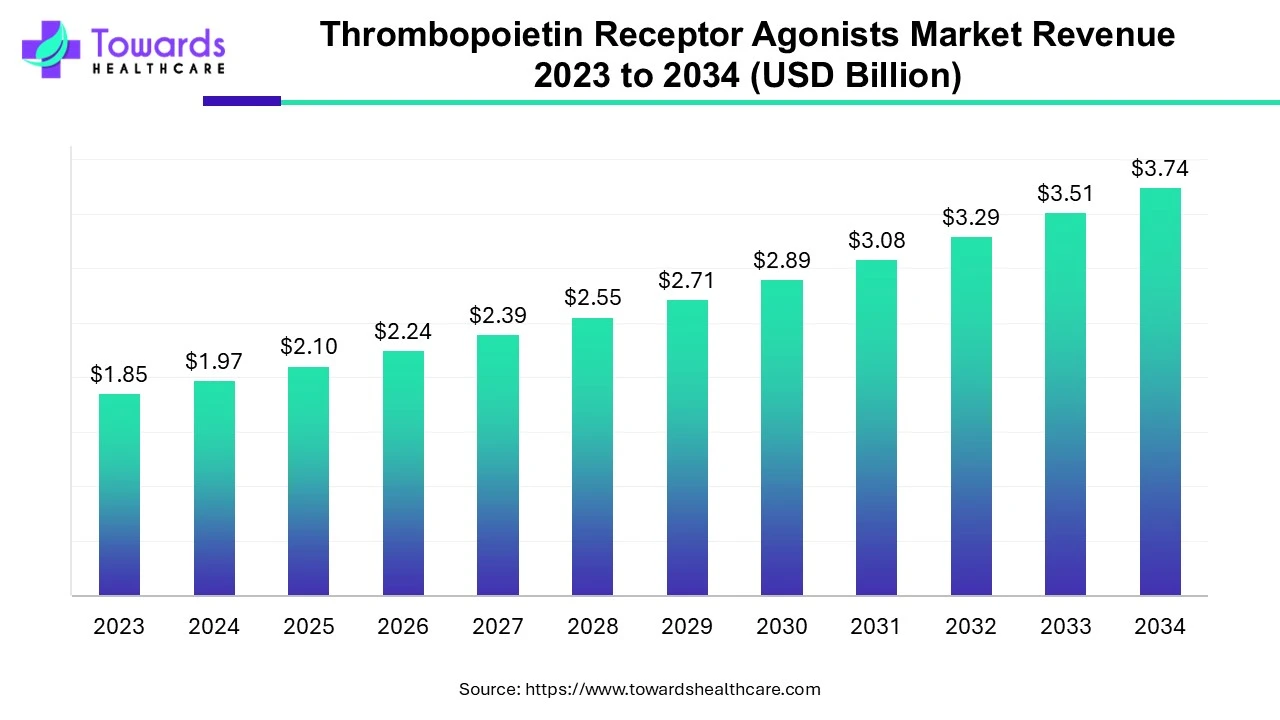
The global thrombopoietin receptor agonists market was estimated at US$ 1.85 billion in 2023 and is projected to grow to US$ 3.74 billion by 2034, rising at a compound annual growth rate (CAGR) of 6.6% from 2024 to 2034. The thrombopoietin receptor agonist drugs are in high demand because they are used for increasing the platelet count, which decreases various health issues.
The global thrombopoietin receptor agonists market is responsible for the production, distribution, and R&D of thrombopoietin receptor agonists (TPO-RAs) drugs in oral or injectable forms. TPO-RAs are used for increasing platelet counts and in preventing bleeding. The U.S. FDA and the European Medicines Agency have approved or not approved different TPO-RAs for different health issues. In the future, in-depth research and knowledge can be gathered on the TPO-RAs class, which will be useful in empowering healthcare professionals and pharmacists to make evidence-based and informed decisions with individual patients, enabling patient-centered care.
One of the challenges faced by the thrombopoietin receptor agonists market is that the drugs are not approved in all countries. The U.S. FDA and the European Medicines Agency have approved certain drugs, but not for all the health issues. This creates a potential growth opportunity for the market. Organizations, government agencies, and research can, individually or in collaboration, conduct research and develop better TPO-RA drugs in order to get approval from governments. Due to this, the drugs will be used in more countries and for a variety of health conditions.
There are certain adverse reactions caused by the use of TPO-RAs, which is why there are many restrictions on their use. These restrictions create challenges in the market’s growth. Some of the adverse effects of using TPO-RAs are rebound thrombocytopenia, thrombocytosis, thrombosis & thromboembolic complications, increase in bone marrow reticulin, antibody formation, cataracts, and transaminase elevation.
North America’s dominance reflects its actions in developing advanced healthcare systems, research and development for better therapeutics, actions taken to increase the reach of healthcare services, and the presence of market key players. Countries like the U.S. and Canada contribute significantly to the market’s growth. The U.S. dominated the thrombopoietin receptor agonists market in North America in 2023, followed by Canada. The countries are making efforts to improve therapeutics due to the growing concern of chronic liver disease and cancer cases.
According to the American Cancer Society, there will be around 20,800 new cases of acute myeloid leukemia (AML) in the United States in 2024. Adults will have the majority of instances. An estimated 11,220 individuals will pass away with AML. Once more, adults will account for the majority of these deaths. The Government of Canada anticipated that 3,100 leukemia fatalities and 6,700 new leukemia cases will occur in Canada (excluding Quebec) in 2022. 77% of individuals with acute myeloid leukemia in Canada (not including Quebec) are expected to pass away within five years after receiving their diagnosis.
The thrombopoietin receptor agonists market is growing due to growing research and development in cancer and other health issues that require TPO-RA drugs. Apart from this, the population in the region is also growing significantly, which increases the manpower or number of patients. This increases the interest of the organizations and the governments to take major steps towards the improvement of the healthcare and pharmaceutical industry.
Europe is considered to be a significantly growing area in the thrombopietin receptor agonists market. The rising prevalence of hematological disorders and growing research and development activities promote market growth. Favorable regulatory policies enable key players to develop novel and innovative TPO-RAs. As of 2023, three TPO-RAs have been approved in Europe. The increasing investments and collaborations among key players also contribute to market growth. The Orphanet database estimated a prevalence of ITP of 1 to 5 per 10,000 and an annual incidence of 1 in 25,600 to 37,000 in Europe.
For instance,
By indication, the acute myeloid leukemia (AML) segment is anticipated to grow rapidly in the market during the forecast period. Induction and consolidation chemotherapy are highly myelosuppressive for acute myeloid leukemia (AML), and CIT is a common burden. A TPO-RA can be used safely and effectively in this case to reduce the risk of bleeding and the need for platelet transfusion. The market is anticipated to grow as the number of leukemia patients increases because more TPO-RA drugs will be used to treat the cancer.
By indication, the immune thrombocytopenia (ITP) segment is estimated to grow at a significant rate during the predicted timeframe. Second-line therapies for ITP include rituximab, splenectomy, and TPO-RAs. Thrombopoietin-receptor agonists (TPO-RAs) are currently the most sophisticated therapy for immunological thrombocytopenia. The findings of this study provide credence to the use of TPO-RAs as second-line therapy for chronic and persistent ITP patients who do not respond to or relapse following previous first-line ITP treatments. TPO-RAs are safe and efficient medications for the treatment of primary ITP.
By dosage form, the injectables segment held the largest share of the market. Injectables have several benefits, which is why they are used by professionals for treatment. The dose is given via injectables, which reduces the time of reaction, and can be given directly to the site of action.
By dosage form, the oral segment is anticipated to grow at the fastest rate during the forecast period. Oral TPO-RA medications are gaining popularity in the healthcare industry. Oral medications do not require the presence of professionals and can be taken by the patients directly. Oral medicine also reduces the cost of the medication.
By end-use, the hospitals segment held the major share of the market, and the segment is expected to grow at the fastest rate during the forecast period. Patients with CLG, AML, ITP, and other health issues are admitted to hospitals for treatment and health monitoring. Due to this, TPO-RAs are used mainly in hospitals. Apart from this, hospitals have all the resources for providing multidisciplinary care, due to which patients prefer hospitals over other healthcare facilities.
Luis Meillón, Hematologist, ABC Medical Center in Mexico City, commented that the diversity of ITP makes it difficult to predict individual responses to treatments, and not all patients achieve effective outcomes. Fostamatinib’s approval offers physicians an innovative therapeutic alternative.

By Indication
By Dosage Form
By End-user
By Region