
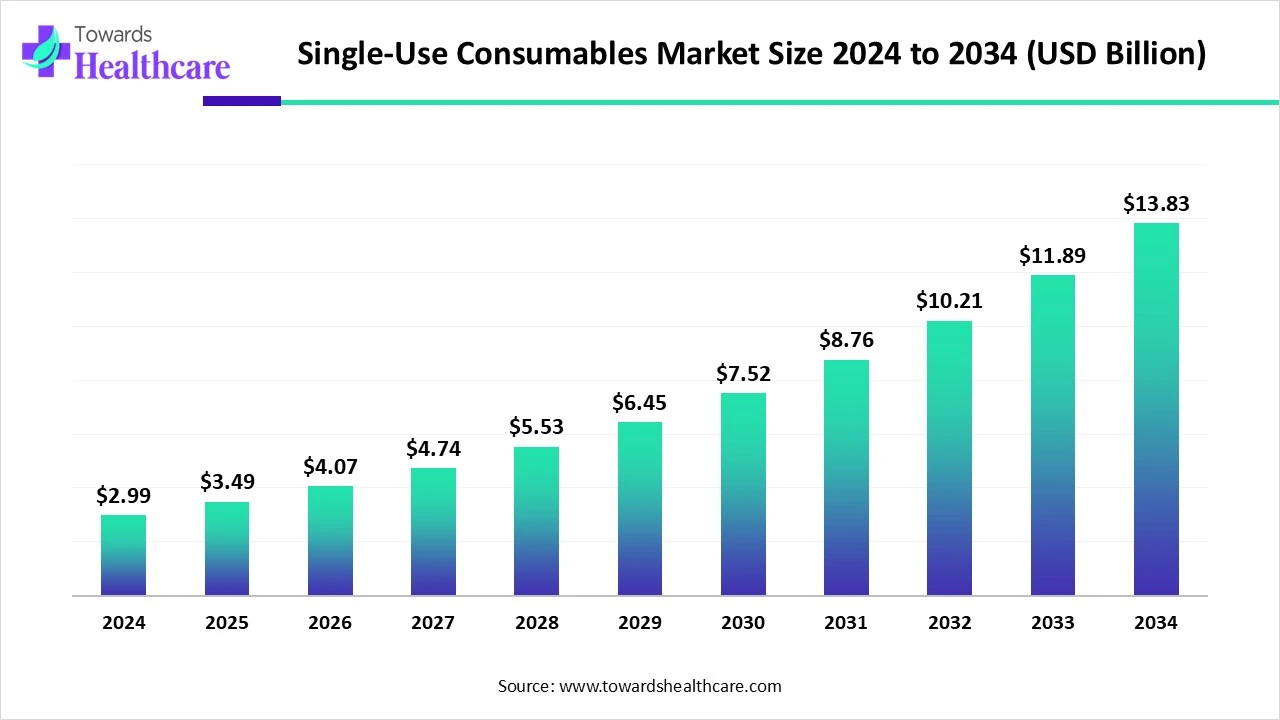
The global single-use consumables market size began at US$ 2.99 billion in 2024 and is forecast to rise to US$ 3.49 billion by 2025. By the end of 2034, it is expected to surpass US$ 13.83 billion, growing steadily at a CAGR of 16.56%.
The single-use consumables market is witnessing substantial growth driven by increasing demand across healthcare, pharmaceutical, and laboratory sectors. Rising awareness about hygiene, contamination prevention, and infection control has intensified the adoption of disposable products such as syringes, gloves, masks, petri dishes, and labware. Technological advancements in materials, including biodegradable and sustainable alternatives, are further boosting market acceptance. The single-use consumables market is witnessing significant growth, led by the dominance of North America, driven by its advanced healthcare infrastructure, high healthcare infrastructure, high healthcare expenditure, and strict regulatory standards.
| Table | Scope |
| Market Size in 2025 | USD 3.49 Billion |
| Projected Market Size in 2034 | USD 13.83 Billion |
| CAGR (2025 - 2034) | 16.56% |
| Leading Region | North America |
| Market Segmentation | By Product Category, By Material / Technology, By Sterility / Use Case, By End User / Care Setting, By Distribution Channel, By Commercial Model / Pricing, By Region |
| Top Key Players | 3M, Medline Industries, Cardinal Health, Becton, Dickinson & Co. (BD), Baxter International, B. Braun Melsungen, Kimberly-Clark Health / Kimtech, Ansell, Terumo Corporation, Smiths Medical, ConvaTec, Coloplast, Mölnlycke Health Care, Owens & Minor, Henry Schein, Fresenius Kabi, Teleflex, Cook Medical, Nipro Medical |
Single-use consumables in the medical field refer to disposable products designed for one-time use to ensure patient safety, sterility, and infection control. These items eliminate the risk of cross-contamination and are widely used in hospitals, clinics, and laboratories. Common examples include syringes, needles, gloves, masks, IV sets, catheters, blood collection tubes, and surgical drapes. Their adoption has grown significantly due to the increasing focus on infection prevention, cost efficiency, and compliance with strict healthcare regulations. Moreover, the rising prevalence of chronic diseases, higher surgical volumes, and advancements in sterile barrier systems are further driving the demand for medical single-use consumables.
The Single-Use Consumables Market comprises single-patient, disposable medical products used across clinical workflows to support patient care, infection control, diagnostics, surgery and outpatient procedures. Examples include syringes & needles, IV/infusion sets, catheters, surgical drapes & gowns, masks, gloves, specimen collection kits, diagnostic swabs, single-use endoscopes/accessories, and single-use device components. Drivers include infection-prevention mandates, regulatory pressure to reduce cross-contamination, one-time-use workflow preferences (especially after high-profile reprocessing safety issues), growth of outpatient/ambulatory care, price competition with reusable alternatives (including total-cost-of-ownership comparisons), and supply-chain/regulatory dynamics that favour reliable single-use supply.
Acquisition or partnering with regional players allows large firms to gain immediate market access and distribution networks without starting from scratch. Adoption of growth strategies even assists in accelerating the manufacturing capacity, innovation of new products, and expansion of business.
Investment and funding drive the growth of the single-use consumables market by enabling product innovation, scaling manufacturing, and expanding distribution. They support the development of sustainable alternatives, automation, and regulatory compliance. Capital enhances brand differentiation, marketing, and supply chain efficiency. Overall, investment accelerates the production, quality, and reach of consumables, helping companies meet rising global demand across sectors like healthcare, food service, and personal care.
For instance,
AI integration can greatly enhance the market by improving efficiency, safety, and sustainability in the medical field. Through predictive demand forecasting, AI helps hospitals anticipate the need for items such as syringes, gloves, and masks, thereby preventing shortages or overstocking. It optimizes supply chains to ensure timely delivery and reduces waste, while machine learning and computer vision strengthen quality control by detecting defects in products like catheters or IV sets in real time. Additionally, AI enables personalized inventory management based on patient inflow, surgical schedules, or disease outbreaks. It also supports sustainability by analyzing waste patterns and suggesting eco-friendly alternatives, while assisting in regulatory compliance to maintain sterility and patient safety standards.
CMOs and CDMOs are Driving the Market
The expansion of contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs) is a key driver of growth in the single-use consumables market, as pharmaceutical and biotechnology companies increasingly outsource biologics, vaccines, and sterile injectable production to reduce costs and accelerate time to market. Single-use systems are particularly valuable for CMOs because they enable rapid scale-up, flexible batch production, and quick changeovers between different products while minimizing downtime and lowering the risk of cross-contamination. Their disposability also reduces the need for costly cleaning validations, making them well-suited for multiproduct facilities and small-batch, high-potency manufacturing such as cell and gene therapies.
In 2025, Akums Drugs & Pharmaceuticals Ltd., a leading Indian CDMO, which commissioned a new sterile manufacturing facility dedicated to lyophilized and injectable products. Since such facilities require stringent sterility, adaptability, and contamination-free production environments, the adoption of single-use consumables is critical to their operation. This instance highlights how the continued expansion of CMOs worldwide is directly fueling the demand for single-use technologies across the pharmaceutical manufacturing landscape.
Environmental Impact & High Disposal Costs
The key players operating in the market are facing issues due to high disposal costs and environmental impact, which is estimated to restrict the market growth. Several factors restrict the growth of the single-use consumables market, particularly in the medical field. The primary challenge is the environmental impact associated with the large volume of plastic and non-biodegradable waste generated, which has led to stricter regulations and rising pressure for sustainable alternatives. Additionally, high disposal costs and the burden of medical waste management increase operational expenses for healthcare facilities.
Rapid Expansion of Vaccine and Biologics Pipelines
The rapid expansion of vaccine and biologics pipelines is one of the strongest growth drivers for the single-use consumables market, as it demands scalable, sterile, and highly flexible manufacturing solutions. With the rising prevalence of infectious diseases and the growing need for next-generation vaccines such as mRNA, viral vector, and protein-based platforms, pharmaceutical companies are under pressure to accelerate production while maintaining stringent quality standards. Single-use consumables, such as bioreactors, filtration units, tubing assemblies, and disposable bags, enable faster setup, reduced cleaning validation, and minimal risk of cross-contamination, making them ideal for high-throughput vaccine and biologics manufacturing. Additionally, the ability to quickly switch production lines supports the development of multiple vaccine candidates simultaneously, an advantage that became evident during the COVID-19 pandemic and continues to drive adoption in other therapeutic areas.
For instance,
The infection control & PPE segment dominates the single-use consumables market due to the critical role these products play in maintaining hygiene, safety, and compliance in healthcare settings. Rising concerns about hospital-acquired infections (HAIs) and the need to prevent cross-contamination have significantly increased the demand for disposable gloves, masks, gowns, and face shields. The COVID-19 pandemic further accelerated adoption, making PPE an essential part of everyday medical practice. Additionally, strict regulatory guidelines on infection control, growing surgical volumes, and the rising prevalence of infectious diseases reinforce reliance on single-use PPE products. Their cost-effectiveness, convenience, and ability to ensure sterility position this segment as the most essential and widely used in healthcare facilities.
The catheters & vascular access segment is estimated to be the fastest-growing product category in the single-use consumables market due to the rising prevalence of chronic diseases such as cardiovascular disorders, diabetes, and kidney-related conditions that require frequent hospitalization and long-term treatments. Increasing surgical procedures, along with the growing need for intravenous drug administration, dialysis, and chemotherapy, are driving higher consumption of catheters and vascular access devices. Single-use options are preferred for their sterility, safety, and ability to minimize infection risks, particularly bloodstream infections. Technological advancements in catheter design, such as antimicrobial coatings and biocompatible materials, further enhance their adoption. Additionally, the expanding elderly population and higher patient admissions in intensive care units contribute to the segment’s rapid growth.
The single-use plastics & polymers segment dominates the material/ technology segment in the single-use consumables market because of their versatility, cost-effectiveness, and ease of large-scale production. Plastics and polymers such as polypropylene, polyethylene, and PVC are widely used in manufacturing syringes, gloves, IV sets, catheters, and packaging due to their durability, flexibility, and ability to maintain sterility. Their lightweight nature and compatibility with sterilization methods make them ideal for healthcare applications. Moreover, plastics ensure barrier protection against contamination and infection, which is crucial in medical environments. Despite growing environmental concerns, their availability, affordability, and regulatory acceptance have kept them the dominant material choice, with ongoing innovations in biodegradable and recyclable polymers further strengthening their position.
The advanced dressings segment is anticipated to be the fastest-growing material/ technology segment in the single-use consumables market due to the rising incidence of chronic and surgical wounds, including diabetic foot ulcers, pressure ulcers, and post-operative wounds, which require effective healing solutions. Advanced dressings, such as hydrocolloid, hydrogel, alginate, and foam-based products, provide superior moisture balance, faster healing, and reduced infection risks compared to traditional dressings. Increasing demand for minimally invasive and patient-friendly wound care, coupled with the growing elderly population prone to chronic wounds, is further fueling adoption. Additionally, technological advancements like antimicrobial coatings and bioactive dressings, along with greater awareness of advanced wound management in both hospitals and home care settings, are driving rapid growth in this segment.
The sterile surgical & procedural consumables segment dominates the market due to its essential role in maintaining aseptic conditions and preventing infections during medical procedures. This segment includes products such as surgical gloves, gowns, drapes, sutures, scalpels, and other disposable instruments, which are critical for patient safety. Factors driving its dominance include rising surgical volumes, an increase in minimally invasive and complex procedures, and growing awareness of hospital-acquired infection (HAI) prevention. Additionally, stringent regulatory standards and hospital protocols mandate the use of sterile single-use items, ensuring high demand and reinforcing the segment’s position as the most vital in healthcare consumables.
The non-sterile patient care consumables segment is estimated to be the fastest-growing segment in the single-use consumables market due to the increasing focus on hygiene, infection prevention, and patient comfort in hospitals, clinics, and home care settings. This segment includes items such as bed pads, adult diapers, wipes, gowns, and other disposable personal care products. Rising elderly population, growing prevalence of chronic and lifestyle-related diseases, and higher demand for home healthcare services are key growth drivers. Additionally, the convenience, cost-effectiveness, and ease of disposal of non-sterile consumables make them attractive for both healthcare providers and patients, further accelerating adoption in medical and caregiving environments.
The hospitals & acute care segment dominates the single-use consumables market due to the high patient volume, complex procedures, and stringent infection control requirements in these settings. Hospitals and acute care centers perform a large number of surgeries, diagnostic tests, and invasive procedures, all of which demand sterile and disposable products such as gloves, syringes, catheters, surgical drapes, and PPE. The need to prevent hospital-acquired infections (HAIs) and comply with strict regulatory standards further drives reliance on single-use consumables. Additionally, increasing healthcare infrastructure, rising surgical volumes, and the presence of advanced intensive care units (ICUs) contribute to sustained high demand, making hospitals and acute care facilities the dominant end-user segment.
The ambulatory surgery centers & outpatient clinics segment is anticipated to be the fastest-growing segment in the single-use consumables market due to the rising preference for minimally invasive procedures and outpatient care. These facilities handle a growing number of same-day surgeries, diagnostic procedures, and minor treatments, all of which require sterile, disposable products such as gloves, gowns, drapes, syringes, and catheters. Factors driving growth include cost-effectiveness, convenience, and reduced hospital stays, which increase patient throughput. Additionally, the expansion of outpatient healthcare services, increasing awareness of infection control, and adoption of advanced procedural consumables further accelerate the demand for single-use products in ASCs and outpatient clinics.
The direct sales & GPO contracts segment dominates the distribution channel in the market due to its ability to ensure consistent supply, cost efficiency, and streamlined procurement for healthcare facilities. Hospitals, clinics, and surgical centers prefer direct contracts with manufacturers or through GPOs to secure bulk orders of items such as gloves, syringes, catheters, and surgical consumables at negotiated prices. This model reduces inventory shortages, administrative burden, and procurement costs, while providing access to a wide range of high-quality products. Additionally, long-term contracts and established relationships with suppliers help healthcare providers maintain regulatory compliance and guarantee timely delivery, reinforcing the dominance of this distribution channel.
The online/e-commerce & B2B marketplaces segment is estimated to be the fastest-growing distribution channel in the single-use consumables market due to the convenience, accessibility, and efficiency they offer to healthcare providers. Hospitals, clinics, and outpatient centers increasingly rely on online platforms to order consumables such as gloves, syringes, catheters, PPE, and surgical dressings quickly and in smaller or just-in-time quantities. The digitalization of procurement processes, coupled with the ability to compare prices, access product reviews, and track shipments in real time, drives adoption. Additionally, the expansion of B2B marketplaces in emerging regions and the growing acceptance of online channels for bulk medical supplies accelerate growth in this distribution segment.
The per-unit purchase/consumable sales segment dominates in the single-use consumables market because it offers flexibility, cost transparency, and ease of procurement for healthcare providers. Hospitals, clinics, and outpatient centers can purchase items such as syringes, gloves, catheters, and surgical consumables according to actual usage, reducing waste and avoiding unnecessary inventory costs. This model is particularly suitable for high-volume, frequently used products where bulk procurement may not be practical. Additionally, per-unit pricing allows healthcare facilities to manage budgets efficiently, maintain regulatory compliance, and respond quickly to fluctuating patient volumes, reinforcing its position as the dominant commercial model in the single-use consumables market.
The managed inventory/scope-as-a-service & subscription bundles segment is anticipated to be the fastest-growing commercial model/ pricing segment in the market due to its ability to streamline supply management, reduce stockouts, and optimize costs for healthcare providers. Under this model, manufacturers or distributors provide a continuous supply of items such as gloves, syringes, catheters, and surgical consumables based on real-time usage data, often through automated replenishment systems. This ensures just-in-time delivery, minimizes inventory holding costs, and improves operational efficiency. Additionally, subscription bundles offer predictable pricing, simplified procurement, and integrated service agreements, which are particularly attractive to hospitals, outpatient clinics, and ambulatory surgery centers, driving rapid adoption of this flexible and efficient commercial model.
North America dominates the market due to its strong biopharmaceutical industry, advanced research infrastructure, and high adoption of innovative manufacturing technologies. The region is home to leading pharmaceutical and biotechnology companies, as well as a large network of contract manufacturing organizations (CMOs/CDMOs) that rely heavily on single-use systems for flexible, multi-product production. Significant investments in biologics, vaccines, and cell and gene therapies further accelerate demand for disposable solutions that ensure sterility, scalability, and cost efficiency. In addition, favorable regulatory support, rising healthcare expenditure, and the presence of key market players strengthen North America’s leadership in this sector.
The U.S. is the largest market in North America, driven by its strong pharmaceutical and biotechnology sector, presence of leading biopharma companies, and advanced research facilities. The country also has a robust pipeline of biologics, vaccines, and cell and gene therapies, which require flexible, contamination-free production supported by single-use technologies. In addition, a high concentration of CMOs/CDMOs and favorable regulatory support further strengthen the U.S. market.
Canada is steadily expanding its role in the region, supported by government initiatives to boost domestic vaccine and biologics manufacturing. Significant investments in biomanufacturing infrastructure and partnerships with global pharma companies are driving demand for single-use consumables. Canada’s focus on expanding its life sciences sector and enhancing production capacity is positioning it as a growing contributor to regional market growth.
The Asia-Pacific region is expected to grow at the fastest rate in the single-use consumables market due to the rapid expansion of its biopharmaceutical sector, increasing investments in vaccine and biologics manufacturing, and rising demand for cost-effective production technologies. Countries like China, India, South Korea, and Japan are strengthening their biomanufacturing capabilities through government support, international collaborations, and infrastructure development. The region’s large patient population, combined with the growing prevalence of chronic and infectious diseases, is fueling the need for advanced biologics and vaccines, thereby boosting the adoption of single-use technologies.
Additionally, the rising presence of global contract manufacturing organizations, coupled with local CMOs scaling up flexible production facilities, further accelerates growth. Competitive labor costs and expanding R&D activities make the Asia-Pacific highly attractive for future investments.
China is the largest contributor in the region, driven by massive investments in biologics, vaccines, and cell and gene therapy manufacturing. Government initiatives, such as the “Made in China 2025” plan and expanded biotech funding, support the adoption of single-use technologies for scalable, contamination-free production.
India’s growing pharmaceutical and biotech industry, combined with a large domestic patient base and rising vaccine production, is boosting demand for flexible and cost-effective single-use systems. Partnerships with global CMOs and expansions of domestic manufacturing facilities further accelerate growth.
Japan’s well-established biotech and pharmaceutical sector, along with high adoption of advanced manufacturing technologies, contributes to steady market expansion, particularly in monoclonal antibodies and biologics production.
South Korea is emerging as a key hub for biologics and cell therapy manufacturing, with government-backed infrastructure development and R&D initiatives driving single-use consumables adoption.

In May 2025, Lucas Muriello, CFO of USMGC stated that the full-scale opening of the U.S. Medical Glove Company's (USMGC) face mask manufacturing facility is a significant step toward bringing the country's health and safety supply chain back to American territory. With six high-speed lines specifically designed to produce 3-ply disposable face masks, the state-of-the-art facility advances USMGC's dedication to supplying essential medical supplies that are produced in the USA.
By Product Category
By Material / Technology
By Sterility / Use Case
By End User / Care Setting
By Distribution Channel
By Commercial Model / Pricing
By Region